Dimitria Electra Gatzia (University of Akron, University of Antwerp)
[Jump to Dimitria’s response to invited comments]
Abstract: Cognition can influence action. Your belief that it is raining outside, for example, may cause you to reach for the umbrella. Perception can also influence cognition. Seeing that no raindrops are falling, for example, may cause you to think that you don’t need to reach for an umbrella. The question that has fascinated philosophers and cognitive scientists for the past few decades, however, is whether cognition can influence perception. Can, for example, your desire for a rainy day cause you to see, hear, or feel raindrops when you walk outside? More generally, can our cognitive states (such as beliefs, desires or intentions) influence the way we see the external world? In the first part of this paper, I present evidence of top-down modulation in early vision. In the second part of the paper, I make a distinction between two types of top-down modulation. The first pertains to the unconscious visual ‘inferences’ the visual system makes as it ‘chooses’ among many possible representations to arrive at one that we experience as a conscious precept (or back-end effects). The second pertains to the cognitive states of perceivers, which may be used to alter the function the visual system computes (or front-end effects). I use this distinction to argue that evidence for top-down modulation in early vision need not threaten the Cognitive Penetrability Thesis (CIT). Colour vision is used as a case study to show how empirical findings suggesting that colour experience is cognitively penetrated can be better explained without reference to cognitive penetration.
1. Introduction
Cognition can influence action. Your belief that it is raining outside, for example, may cause you to reach for an umbrella. Perception can also influence cognition. Seeing that no raindrops are falling, for example, may cause you to think that you don’t need to reach for an umbrella. The question that has fascinated philosophers and cognitive scientists for the past few decades, however, is whether cognition can influence perception. Can, for example, your desire for a rainy day cause you to see, hear, or feel raindrops when you walk outside? More generally, can our cognitive states (such as beliefs, desires, or intentions) influence the way we see the external world? The goal of this paper is to investigate this question.
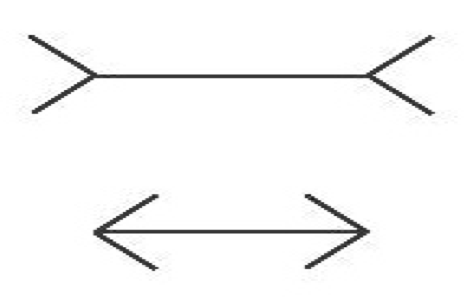
Cognitive penetration (CP) refers to the influence cognitive states, e.g., beliefs, desires, or intentions, have on perceptual experience. In the twentieth century, the possibility of cognitive penetrability in perception was the core tenet behind the New Look movement in psychology, which studied several alleged cases, albeit without appeal to a precise notion of cognitive penetration (Brunner and Goodman, 1947; Brunner and Postman, 1949). In the late eighties, stern criticism from philosophers and psychologists, who were primarily concerned with the characterization of a reliable visual system capable of representing the world accurately, led most to reject the claim that perception is cognitively penetrated (Fodor, 1983; Pylyshyn, 1984; Pylyshyn, 1999). By the late nineties, the Cognitive Impenetrability Thesis (CIT), according to which cognitive states do not influence the content (what the experience is about) or qualitative character (what the experience is like) of experience, had become the dominant view in philosophy. The Müller-Lyer illusion (see Fig. 1) has since served as the quintessential example by proponents of the CIT, as it seems nicely illustrates that acquiring the belief that the lines have the same length cannot influence the perceptual content or qualitative character of one’s experience; one line continues to look shorter than the other even after one learns that they have the same length.
The current has once again changed. Recently, philosophers have used empirical studies to challenge the CIT (Macpherson, 2012; Siegel, 2012; Wu, 2013; Cecchi, 2014; Wu, in press). The debate about the possibility of cognitive penetrability in perception has primarily focused on early vision. Generally, proponents of the CIT allow the top-down modulation of late vision (which consists of high-level processes) but argue that early vision (which is restricted to low-level processes) is impervious to cognitive influences.
In what follows, I begin with the context in which to understand the debate about the CIT (section 2) and show that early vision is subject to top-down modulation (section 3). I argue that a distinction should be made between mere top-down modulation and cognitive penetration. While the former refers to the unconscious visual ‘inferences’ the visual system makes as it ‘chooses’ among many possible representations to arrive at one that we experience as a conscious precept, the latter refers to the purported influences the cognitive states of perceivers, e.g., their beliefs, desires, or intentions, have on their visual experiences (section 3). I use this distinction to argue that evidence for top-down modulation (even in early vision) need not threaten the CIT (section 4). Lastly, I use this distinction to show that empirical findings that have been used to argue that colour experience is cognitively penetrated can better be explained without reference to cognitive penetration (section 5).
2. Visual Perception: top-down vs. bottom-up modulation
Vision is not as straightforward as it appears to the observer. The only available information to the visual system is a given retinal image, which does not provide accurate or complete information about a given external or distal stimulus. Indeed, the mapping from retinal image to distal stimulus is not one-to-one, but rather one-to-many, meaning that identical distal stimuli (under different conditions of illumination or at different distances and orientations from a perceiver, and so on) can give rise to radically different retinal images while radically different distal stimuli can generate the same retinal image. Despite this, the visual system is somehow (we do not yet know the precise mechanisms) able to recover useful information about the environment (Brainard, 2009; Purves and Lotto, 2003).
According to the dominant view in cognitive science, visual information is processed sequentially in a feedforward (or bottom-up) manner: visual inputs from subcortical areas such as the retina and the lateral geniculate nucleus (LGN) are transmitted along the ventral pathway[1] to the primary or striate (V1), pre-striate (V2), and V4 (which shows attentional modulation) cortices before reaching, first, the middle temporal cortex (MT or V5), and, then, the inferior temporal cortex (IT), which consists of the posterior (PIT), central (CIT), and anterior (AIT) cortices (see Fig. 2) (Yasuda et al., 2010).[2]
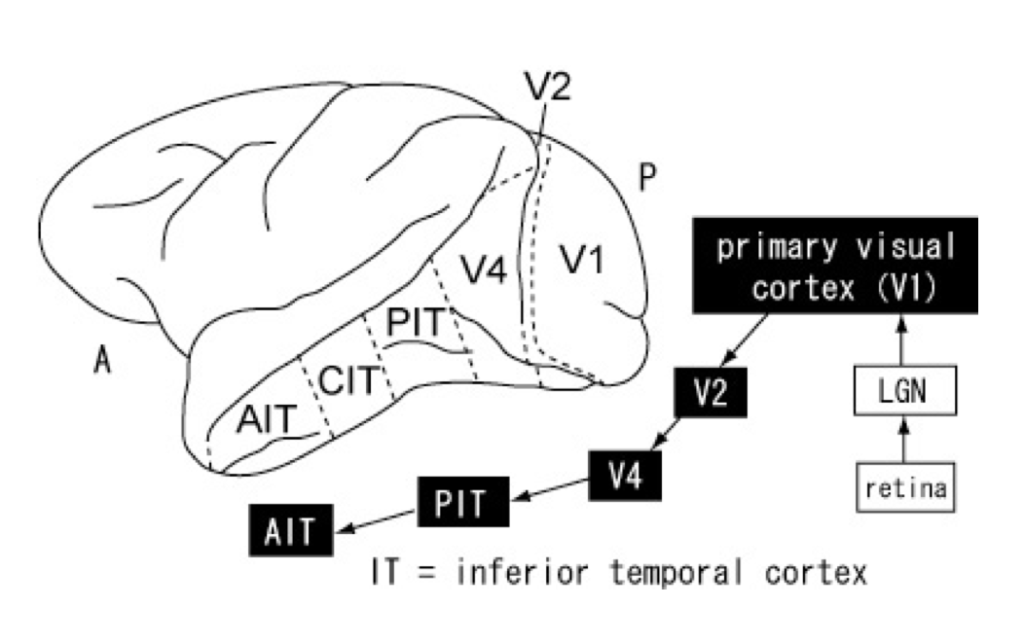
The dominance of the hierarchical (bottom-up) model of visual perception has led to the demarcation of some cortical areas (such as V4 and IT) as ‘higher-level’ to indicate that they involve steps at the top of the processing hierarchy and other cortical as well as subcortical areas (such as V1 and LNG respectively) as ‘lower-level’ to indicate that they involve steps at the bottom of the processing hierarchy. “Bottom-up” describes the direction of information processing: information is processed hierarchically from low-level subcortical and cortical areas to higher-level cortical areas. “Top-down” describes the opposite direction information is processed from higher-level cortical areas to lower-level cortical and subcortical areas.
The question of whether information is processed in a bottom-up or top-down fashion is relevant to questions about the role of visual perception. For example, the view that information is processed in a bottom-up fashion indicates that the visual system is data-driven. This suggests that the role of visual perception is to form (for the most part) accurate representations of the external world (Marr, 1983). The view that information is processed in a top-down fashion, on the other hand, suggests that the visual system is cognition-driven. On this view, far from merely reconstructing visual scenes, the visual system constructs a “hypothesize reality” (Palmer, 1999).[3] This suggests that the role of visual perception is to provide information that is essential in successful behavior.[4]
The debate about the CIT can be placed within the debate about the role of visual perception. Those who adopt the bottom-up model of visual perception tend to deny that visual perception is top-down modulated and embrace the CIT. Those who adopt the top-down model postulate that visual perception is top-down modulated and reject the CIT. In a recent paper Marchi and Newen (2015) argue that experiences of facial expressions are cognitively penetrated by higher-level cognitive states. To support this claim, they present a study by Carroll and Russell (1996) on the relevance of context in the recognition of facial expressions. Subjects were asked to evaluate the photograph of a woman’s face. In the absence of a context, subjects evaluated the face as expressing fear. However, when they were given a context—when subjects were told that a woman who had made reservations for her and her sister at an expensive restaurant is made to wait for over an hour while the maître repeatedly gave available tables to other patrons—they evaluated the same face as expressing anger. After defining cognitive penetration in terms of the presence of an internal and mental causal dependency between a perceptual experience and a cognitive state (Stokes, 2013: 650; for a detailed definition see section 4), Marchi and Newen argue that the best explanation for these results is that subjects had two different experiences of the same face, not that subjects attributed different emotions to the same face by forming two different perceptual judgments. They conclude that the subjects’ perceptual experiences are penetrated by higher-level cognitive states. However, there is another explanation (in addition to the two the authors provide) that Marchi and Newen do not consider, which seems to provide an even better explanation for the results of this study.
Firestone and Scholl (in press) have recently argued that studies attempting to show that visual experience is cognitively penetrated fall prey to various pitfalls. One of these pitfalls is failure to distinguish between response variations resulting from differences in perception and those resulting from differences in judgments. It is not always clear whether it our cognitive states that affect our visual experience or merely the judgments we make on the basis of non-experiential information. Marchi and Newen (2015) take great pains to show that it is not the case that subjects in the above study attribute two different emotions to the same face by forming two different perceptual judgments. However, they neglect to consider another explanation associated with another of the pitfalls discussed by Firestone and Scholl, viz. “memory and recognition.” Specifically, the authors distinguish between two types of effects related to recognitional tasks: ‘front-end’ and ‘back-end’. A front-end effect on visual processing is distinct from the role high-level cognitive states such as memory plays on object recognition. As such it would support the claim that cognitive penetration occurs since such an effect would indicate a top-down modulation effect in perception. A back-end effect on memory access such as activating the relevant representations in memory when a stimulus is present (or even before a stimulus is presented as when subjects who are hungry were found to be better at recognizing hunger words since they are related to what they are already thinking about) would not. Carroll and Russell’s study seems to be a case of back-end effect on memory access: the context activates the relevant representations in memory when a stimulus (in this case the picture of the face) is presented and the subjects respond that they see the face as expressing anger. In the absence of the context, the relevant representations in memory are not activated and the subjects respond that they see the same face as expressing fear. Marchi and Newen do not consider this possibility. And since their argument is an inference to the best explanation, they fail to show that this is a case of cognitive penetration.
This case illustrates that the occurrence of top-down modulation alone is insufficient to establish the occurrence of cognitive penetration. Indeed, “normal” perception is contingent on strong top-down modulation. For example, several studies indicate that schizophrenic subjects are less susceptible than controls to three-dimensional depth inversion illusions such as the hollow-mask illusion, which occur when concave objects appear convex, than healthy controls (Schneider et al., 1996; Schneider et al., 2002; Koethe et al., 2006; Dima et al., 2009; Keane et al., 2013). Using functional magnetic resonance imaging (fMRI), Dima and colleagues (2009) found that poor top-down modulation of the fronto-parietal network in schizophrenic subjects accounted for their diminished susceptibility to the hollow-mask illusion. The fact that schizophrenic subjects exhibit such perceptual abnormalities indicates that “normal” vision depends on strong top-down modulation; for, although poorer top-down modulation would give us an advantage in terms of reduced susceptibility to illusions, it would also decrease our ability to effectively interact with our environment. Indeed, studies show that subjects who recovered from prolonged blindness are also less susceptible to depth illusions (Kurson, 2007; Gregory 2004, 2009). This advantage, however, comes at a high cost: they are also unable to recognize familiar objects when they see them (possibly because their visual system is unable to activate the relevant representations in memory when a stimulus is present).[5] What is needed is an argument that top-down modulation involves the perceiver’s cognitive states (as opposed to high-level information processing systems of the visual system). For example, it requires showing that there is a front-end effect on visual perception itself. This point has been lost in the recent debate about cognitive penetration. Even proponents of the CIT who acknowledge the occurrence of top-down modulation of visual perception have devoted their resources to establishing that “early vision” is not subject to top-down modulation. In what follows, I briefly show that even early vision is subject to top-down modulation and argue that there is no need for concern because the presence of top-down modulation on early vision need not threaten the CIT.
3. Top-down modulation in early vision
Theorists who frame the issue of the possibility of cognitive penetrability in terms of top-down modulation, by end large, do not deny the presence of top-down modulation in late vision. However, they argue that these findings do not threaten the CIT because top-down modulation does not extend to early vision (Pylyshyn, 1999; Raftopoulos, 2001). Marr (1976) was the first to use the term “early vision” to refer to early stages of visual cortical processing. Although he initially defined “early vision” functionally, as the first level of visual processing, for which he coined the term primal sketch, he later identified the primary visual cortex (V1) as the neuroanatomical area in which the primal sketch is constructed (see Fig. 2).[6] Unlike Marr, Pylyshyn (1999: 344) is reluctant to locate early vision neuroanatomically noting that its neuroanatomical locus is not yet known with any precision. He too defines “early vision” functionally, but in terms of its psychophysical properties, which include “a mapping of various substages involved in computing stereo, motion, size, and lightness constancies, as well as the role of attention and learning” (Pylyshyn, 1999: 344). Raftopoulos (2009: Ch. 2) also defines “early vision” functionally, but unlike Pylyshyn, in terms of response latency: as the process lasting no more than 100-120 milliseconds (ms) post-stimulus onset; it is a pre-attentional stage, which includes lateral and recurrent processes devoid of cognitive signals.[7]
Marr saw the primary visual cortex (V1) as a low-level processing module where local features such as oriented edges or bars from the proximal image are extracted before being transmitted to higher-level visual areas for further processing (Marr, 1982; see also Hubel and Wiesel, 1978). Although Marr (1976) acknowledged that not all neural computations are feedforward (or bottom-up), he maintained that early visual stages are completed independently of later stages.[8] However, contrary to Marr, empirical findings suggest that V1 (a low-level cortical area) plays a central role in integrating and coordinating computations amongst the higher-level visual areas utilizing the recurrent network connections in the visual system (Deco and Lee, 2004; Mumford, 1996; Lee et al., 1998). For example, studies show that higher-level computations that involve high-resolution details such as fine geometry and special precision take place in V1 and are reflected in its neural activities that encode orientation and spatial information (Lee et al., 1998; Deco and Lee, 2004; Gilbert and Li, 2013.). In addition, empirical data suggests that low-level computations cannot be completed prior to the commencement of high-level computations (Lee et al., 1998). For example, figure-ground separation (associated with low-level computations) and object recognition (associated with high-level computations) are interwoven and cannot progress in a simple feedforward fashion, but have to occur concurrently and interactively in constant feedforward and feedback loops that involve the entire circuit in the visual system (Pollen, 1999; Rao et al., 1999; Lee et al., 1998; Dayan et al., 1995). Top-down modulation is, in fact, seen at all stages of visual processing, including subcortical areas such as the LGN (O’Connor, et al., 2002; McAlonan et al., 2008). The source of top-down influences can be ubiquitous either in the form of direct connections from different cortical areas or as a cascade of inputs originating from different areas (Gilbert and Li, 2013; see Fig. 3).
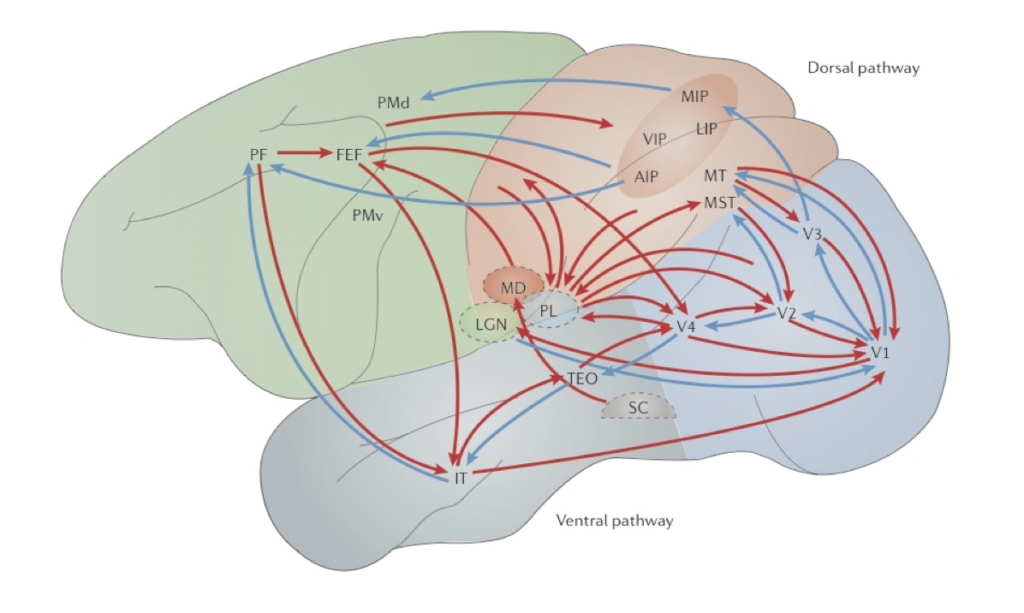
Pylyshyn (1999) acknowledges that complex computations are carried out in early vision. For example, he maintains that cases of perceptual filling-in that appear to involve top-down processing suggest that the “interpretation of parts of a stimulus may depend on the joint (or even prior) interpretation of other parts of the stimulus, resulting in global-to-local influences such as those studied by Gestalt psychologists,” indicating that early vision may embody some local vision-specific memory (Pylyshyn, 1999: 344). However, he argues that that vision is influenced by cognition at only two loci: (a) in the allocation of attention to certain locations or certain properties prior to the operation of early vision; and (b) in the decisions involved in recognizing and identifying patterns after the operation of early vision (Pylyshyn, 1999: 344). In other words, top-down modulation occurs but only pre- or post-perceptually. Pylyshyn uses this distinction to argue that many apparent examples of cognitive penetration arise either from pre-perceptual (attention-allocation) processes or post-perceptual (decision) processes. Since the outcome of visual perception involves either pre-perceptual or post-perceptual processes, Pylyshyn denies that the aforementioned cases (e.g., cases of perceptual filling-in) count as cases of cognitive penetration. However, empirical studies suggest that the interaction of information from the ventral and dorsal pathways can occur in early vision and that the recurrent interaction between higher-level cortical areas and early visual areas such as V1 and V2 seem to play an important role in mediating visual search and attentional routing (Deco and Lee, 2004). Of course, since Pylyshyn does not locate early vision neuroanatomically, it is not clear whether, or to what extend, these findings threaten his claim that the outcome of visual perception involves either pre-perceptual or post-perceptual processes. Nevertheless, there are reasons for doubting the view of early vision as a pre-attentional stage. For example, studies show that the binding of features such as colour, shape, texture, associated with distinct sense modalities can occur in just 40-60 ms (Schoenfeld et al., 2003). Since attention (including object-based selection and, perhaps, attention to locations) plays a crucial role in feature binding (Worden and Foxe, 2003), these findings suggest that early vision is not a pre-attentional stage.
Raftopoulos (2011 and 2009: Ch. 2) also allows for the possibility of top-down modulation of late vision. He too, however, maintains that early vision, which he defines as the processing stage that lasts no more than 100-120 ms post-stimulus onset, is impervious to top-down modulation.[9] Nevertheless, studies show that the transmission rates through the visual cortex are remarkably rapid. Virtually the entire visual system (including higher-level cortical areas such as the IT) becomes activated within 30 ms of the initial afferent input to area V1 (Schroeder et al., 1998). These studies indicate that 100-120 ms is actually a remarkably long time in terms of neural transmission, and that many regions of the cortex can be activated in half that rate (i.e., within 50 ms post-stimulus onset). In fact, studies on humans indicate that even regions of the frontal cortices (associated with cognitive functions such as thinking, problem-solving, and emotions) are activated by visual stimuli within 30-40 ms of initial V1 activation, at a latency of only 70–90 ms post-stimulus onset (Foxe and Simpson, 2002). Robust contextual modulation was also found when disparity, colour, luminance, and orientation cues variously defined a textured figure centered on the receptive field of neurons in area V1 with a characteristic latency of 80-100 ms after stimulus onset, suggesting feedback influences from extrastriate areas (Zipser et al., 1996). These findings suggest that feedback processes impact the ongoing visual processing (Foxe and Simpson, 2002) and, therefore, challenge Raftopoulos’ claim that during the first 100-120 ms post-stimulus onset early vision is impervious to top-down modulation. What all these findings show is that if cognitive penetration is understood in terms of top-down modulation of early vision, it is not difficult to refute the CIT.
4. Top-down modulation vs. cognitive penetration
The Cognitive Penetrability Thesis (CIT) has been traditionally formulated as a semantic thesis, according which the function a system computes is not sensitive (in a semantically-coherent way) to the perceiver’s cognitive states and cannot be altered in a way that bears some logical relation to them (Pylyshyn, 1999: 343; Raftopoulos, 2001; Brogaard and Gatzia, 2015).[10] Semantic coherence refers to a rough correspondence between the content of the perceiver’s cognitive states and the content of her visual experience. For example, suppose that you are not susceptible to the Müller-Lyer illusion (say, because you are suffering from schizophrenia) and as a result you experience the lines as having the same length. All things being equal, your belief (that the lines have the same length) and your visual experience (of the lines as having the same length) are semantically coherent—that is, they have roughly the same content. Now suppose that you are susceptible to the Müller-Lyer illusion.[11] All things being equal, your belief that the lines are the same length differs from your visual experience since you continue to experience one line as being longer than the other even after you learn that they have the same length. Your belief (that the lines have the same length) and your visual experience (of the lines as having varying lengths) are thus said to be semantically incoherent—that is, they do not have the same content.
What is most relevant in the debate about CP understood as a semantic thesis is that your belief (that the lines have the same length) cannot alter the function your visual system computes in a way that it can alter the output (i.e., your visual experience). That explains why you continue to experience the lines as having varying lengths even after you acquire the belief that they have the same length. In order for your belief (that the lines are the same length) would have to alter the function of your visual system in such as way as alter the output (you visually experience the lines as having the same length). However, that does not happen. It follows that acquiring the belief that the lines are of the same lengths is insufficient for altering a visual experience. Showing that cognitive penetration occurs, therefore, requires showing that the changes the visual system undergoes are somehow tied to the perceiver’s cognitive states (in a semantically-coherent way). This distinction is often lost in the literature. For example, Stokes (2013: 650) defines CP as follows:
(CP) A perceptual experience E is cognitively penetrated if and only if (1) E is causally dependent on some cognitive state C and (2) the causal link between E and C is internal and mental.
This definition fails to distinguish between cases of top-down modulation described above associated with back-end effects and cases of cognitive penetration (associated with front-end effects) since all that it requires is that the causal link between the experience and the cognitive state is internal (i.e., in the head). Indeed, as I mentioned earlier, this is the definition that Marchi and Newen (2015) use to arrive to the conclusion that experiences of facial expressions are cognitively penetrated by higher-level states. This definition suggests that rejecting the CIT is a matter of pointing to an internal causal link between a perceptual experience and a higher-level state. Although Marchi and Newen show that there is an internal causal link between a perceptual experience and higher-level state (i.e., a back-end effect on memory access), they fail to show that this is a case of cognitive penetration. A better definition is thus needed. Formulating CP as a semantic thesis (see above) allows us to distinguish between top-down modulation (typically, associated with the operations within the visual system, whose functions cannot be altered by the perceiver’s cognitive states such as beliefs) and cognitive penetration (which requires that the function the visual system computes is altered by the perceiver’s cognitive states, i.e., her beliefs, desires, or intentions).
The presence of top-down modulation merely shows that vision “can have a independent existence with extraordinarily sophisticated inferences that are totally separate from standard, everyday, reportable cognition” (Cavanagh, 2011: 1540). For example, empirical studies indicate that the knowledge of colour-luminance relationships between the aligned chromatic and luminance variations is build into the machinery of the human visual system (see Kingdom, 2003).[12] This ‘knowledge,’ however, has an independent existence (i.e., it is not accessible to the perceiver) with extraordinarily sophisticated ‘inferences’ (i.e., computations used by the visual system). Evidence for top-down modulation seem to support the claim that vision has its own ‘inference’ mechanisms which cannot be altered by the perceiver’s cognitive states. In other words, they support the claim that vision is not a purely bottom-up process. However, they do not support the claim that a perceiver’s cognitive states can penetrate her visual experience. As Cavanagh (2011: 1539) notes,
Saying that the components of high-level vision are the contents of our visual awareness does not mean that these mental states are computed consciously. It only means that the end point, the product of a whole lot of pre-conscious visual computation is an awareness of the object or intention or connectedness. In fact, what interests us here is specifically the unconscious computation underlying these products, and not the further goal-related activities that are based on them… We are interested in the rapid, unconscious visual processes that choose among many possible representations to come up the one that we experience as a conscious percept. Attention and awareness may limit how much unconscious inference we can manage and what it will be focused on but it is the unconscious decision processes that are the wheelhouse of visual cognition.
The visual system transforms sensory inputs into “solid visual experiences with little or no evidence of the inferences that underlie it” (Cavanagh, 2011: 1539). The occurrence of perceptual errors (e.g., the Müller-Lyer) may alert us of the existence of these operations that underlie the visual system but cannot grant us access to them (see, for example, section 5). A distinction, therefore, must be made between the ‘inferences’ the visual system makes as it ‘chooses’ among many possible representations to arrive at one that we experience as a conscious precept, which are not consciously accessible to the perceiver, on the one hand, and the cognitive states of perceivers (i.e., their beliefs, desires, intentions, which are consciously accessible to the them) which may be able to alter the function their visual system computes, on the other. This distinction is implicit in Fodor’s (1988: 194) response to Churchland (even though it relates to the question of whether the brain’s plasticity should be viewed as a case of diachronic cognitive penetration):
What Churchland needs to show—and doesn’t—is that you also find perceptual plasticity where you wouldn’t expect it on specific ecological grounds; for example, that you can somehow reshape the perceptual field by learning physics.
Fodor’s point here is that changes to visual perception that result from the recalibration of the perceptual/motor mechanisms (such as hand/eye) that correlate bodily gestures with perceived spatial positions and which are required for an organism to grow do not count as cases of cognitive penetration because they are adaptational or developmental. As such they are not brought about by the perceivers’ beliefs, desires, or intentions—indeed, they could not have been brought about by learning physics; they are associated with adaptations of the visual system, which result from changes to the environment. To see how this criticism applies to the issue of cognitive penetration let us take a look at purported counterexamples to the CIT.
5. The effects of colour constancy on colour perception
Colour experience is an interesting case for two reasons. The first is that it has been traditionally considered to be cognitively impenetrable, even by those who reject the CIT. The second is that empirical studies have been recently used to challenge the CIT (Delk and Delk and Fillenbaum, 1965; Hansel et al., 2006; Olkkonen et al., 2008).[13] I discuss two such studies (an older and a newer one) and argue that once the above distinction between mere top-down modulation and cognitive penetration is made they lose their force.
In an older study, Delk and Fillenbaum (1965) cut 10 figures out of the same cardboard that had a orange-red colour approximating Muncell chip R/5/12. Some of the figures represented objects that have characteristically red colours (such as apples or a pair of lips) while others represented neutral figures (such as mushrooms or geometric shapes). Subjects were asked to match the colour of each figure, shown to them separately, with a background colour using a differential colour-mixer, which allowed the mixture of two colours. These two colours could be varied from a red shade approximating Munsell chip R/3/8 to a yellow-orange shade approximating Munsell chip YR/6/10—to produce a continuously varying intermediate shade (Delk and Fillenbaum, 1965: 291). Both the figure and the background were illuminated by a fluorescent lamp, which provided a soft-illumination of both figure and ground and cast no shadows on either. They found that subjects adjusted the background colour to a red hue when matching it with figures representing characteristically red objects (such as pair of lips or apple) but to a reddish-orange hue when matching it with figures representing neutral objects (i.e., objects that are not characteristically red such as mushrooms or geometric shapes).
In a more recent study, Hansen and colleagues (2006) presented subjects with either digitized photographs of natural fruit (such as bananas) or random patches (such as geometric shapes) each placed against a gray background. They asked subjects to adjust the colour of the digitized photograph (either a fruit or a random patch) until it appeared gray. They found that subjects adjusted the colour of the banana, but not the random noise patches, to a slightly bluish hue (the opposite of yellow).
Macpherson (2012) argues that Delk and Fillenbaum’s study provides a counterexample to the CIT. Like Marchi and Newen (2015), Macpherson argues that the best explanation for these results is that subjects experienced the cutouts of characteristically red objects as being redder than the controls, not that subjects formed two different perceptual judgments. She thus concludes that the subjects’ perceptual experiences were penetrated by their beliefs about the colours of familiar objects. A similar argument could be made about the study by Hansen and colleagues: subjects experienced the banana as being less gray than the controls, not that subjects formed two different perceptual judgments. Although it is tempting to view these studies as evidence against the CIT, their results are best explained by reference to colour constancy mechanisms, whose computations cannot be altered by the perceiver’s cognitive states.[14]
There are two types of colour constancy: simultaneous and successive (Ling and Hurlbert, 2007).[15] Simultaneous colour constancy occurs when perceivers experience identical surface materials in a scene to have the same colour, even though they are illuminated differentially. For example, if a part of a single surface is directly lit while the other part is in shadow (that is, lit only via the ambient illumination) the entire surface would look to have the same colour. The visual system ‘discounts’ the spatial change in illumination and recovers a constant surface reflectance across the shadow border. A recent failure in successive constancy involving such an effect recently shocked the public (and prompted a plethora of news articles from respectable news outlets) when a photograph of a blue-black dress was posted online (see Fig. 9). What was peculiar about this digitized photograph is that while the dress looked blue and black to some, it looked white and gold to others.

This peculiarity can be explained in terms of simultaneous colour constancy. One part of the dress was directly lit while the other part was in shadow, and lit only via the ambient illumination. As a result, it conferred inconsistent cues to the visual systems of different perceivers. In order to resolve ambiguities arising from contradictory inputs, the visual system discounts the spatial change in illumination and recovers a constant surface reflectance across the shadow border. So although the entire dress looks to have constant colours, some perceivers experience it as blue-black while others as white-gold. When the visual system interprets the scene as being illuminated by a blue light (which is the illumination the visual system typically encounters in natural daylight), the dress looks white-gold (because it discounted the blue light, see image A in Fig. 5). But when the visual system interpreted the scene as being lit by a yellowish light (which is the illumination the visual system encounters in the presence of a shadow), the dress looks blue-black (see image C in Fig. 5). In either case, perceivers would be unable to alter the colour constancy computations their visual systems performed (and which resulted in radically different experiences) even if they were aware of them. Indeed not only they were surprised to find out that the dress looked to have different colours to other people, but also this realization had absolutely no effect on their visual experiences: their colour experiences remained unchanged.

Successive colour constancy occurs when we see the same object as having the same colour under different illuminations. Although the colour of the object undergoes (in many cases radical) changes under different illuminations (see, for examples, the series of paintings by Monet), successive colour constancy mechanisms ensure that (for the most part) these changes go unnoticed. The results from Delk and Fillenbaum’s (1965) study can be explained in terms of successive colour constancy.[16] Recall that subjects adjusted the background to a redder hue when matched it to the characteristically red figures but to a reddish-orange hue when matched it to the controls. This seems to be a case of a back-end effect on memory access where the relevant representations in memory are activated when a stimulus is present. In this case, the relevant representations in memory may have been activated when the characteristically red stimuli were presented (in this case the apple or the pair of lips) but not when the controls were present. This would explain why subjects adjusted the background to a red hue but not the controls: the relevant representations in memory are only activated for the characteristically red objects but not the controls making the visual system prone to error. However, these results do not support the claim that cognitive penetration occurred since these subjects would be unable to alter computations they have no conscious awareness of in such a way as to alter their visual experience. So although the above explanation makes reference to top-down modulation (i.e., to back-end effects), they do not provide a good reason for rejecting the CIT.
The same can be said about the study by Hansen and colleagues (2006). In this case, subjects adjusted the colour of the banana (but not the random patches) to a slightly a bluish hue—the opposite of yellow—in attempting to make it appear gray. Accordingly, subjects adjusted the banana to a slightly bluish hue (the opposite of yellow) but not the random patches. This seems to be a case of a back-end effect on memory access where the relevant representations in memory are activated when a stimulus is present. In this case, the relevant representations in memory may have been activated when the characteristically yellow stimuli were presented (in this case the banana) but not when the random patches were present. This would explain why subjects adjusted the banana to a slightly bluish hue but not the random patches: the relevant representations in memory are only activated for the characteristically yellow objects but not the random patches making the visual system prone to error. Certainly, Hansen et al. (2006: 1368) seem to find this explanation plausible:
Another question posed by our results is whether they are applicable to the type of lighting conditions occurring naturally. Notably, natural illumination during the course of the day varies mostly along the blue-yellow dimension. Natural daylight contains a high proportion of short-wavelength energy in the morning and gradually shifts toward energy of longer wavelengths during the course of the day. Most fruit objects are yellowish rather than bluish; therefore, they might appear grayish in the morning, as color constancy is known to be imperfect even in realistic situations. The appearance shifts described here may contribute toward color constancy under these conditions (emphasis added).
However, these results do not support the claim that cognitive penetration occurred since these subjects would be unable to alter computations they have no conscious awareness of in such a way as to alter their visual experience. So although the above explanation makes reference to top-down modulation (i.e., to back-end effects), they do not provide a good reason for rejecting the CIT. This conclusion is independently supported by the fact that although colour constancy plays an important role in object recognition (e.g., when the visual system retrieves relevant memory representations when a stimulus is present), colour constancy computations are not obligatorily linked to experiencing objects and may precede the experiencing of objects (Kentridge et al., 2014). Given that these mechanisms are not obligatorily linked to experiencing objects and may precede the experiencing of objects, any changes to the function a visual system computes, which may alter one’s colour experience, cannot be attributed to the perceiver’s beliefs, desires, or intentions. It follows that the findings of the aforementioned studies cannot be used as evidence against the CIT.
6. Conclusion
In section 2, I provided the context in which to understand the debate about the CIT. In section 3, I showed that early vision is subject to top-down modulation. In section 4, I argued that once we distinguish between top-down modulation resulting from the computations performed by the visual system (i.e., back-end effects), which cannot be altered by the cognitive states of perceivers since they are not consciously accessible to them and cognitive penetration (i.e., front-end effects), it becomes clear that that evidence for top-down modulation (even in early vision) need not threaten the CIT. In the last section (5), I used this distinction to show that empirical findings that have been used to argue that colour experience is cognitively penetrated can better be explained by reference to back-end effects, specifically, memory and recognition (section 5).
References
Arnsten, A. (2009). Stress signaling pathways that impair prefrontal cortex structure and function. National Review of Neuroscience 10(6): 410-422.
Bartleson, C. J. (1960) Memory Colors of Familiar Objects. Journal of the Optical Society of America 50(1): 73-77.
Blumenfeld, R. S., Ranganath, C. (2007). Prefrontal cortex and long-term memory encoding: an integrative review of findings from neuropsychology and neuroimaging. The Neuroscientist 13(3): 280-91.
Borenstein, E., Ullman, S. (2008). Combined top-down/bottom-up segmentation. IEEE Transactions on Pattern Analysis and Machine Intelligence 30(12): 2109-2125.
Brogaard, B. and Gatzia, D. E. (In press) Color and Cognitive Penetrability. Topics in Cognitive Science: Special Issue on Cortical Color.
Bruce, V. and Burton, M. (2002). Learning to recognize faces. In M. Fahle and Poggio T. (Eds.) Perceptual Learning, pp. 317-334. Cambridge (MA): MIT Press.
Brunner, J. S. and Goodman, C. C. (1947) Value and need as organizing factors of perception. J. Exp. Psychol. Hum. Percept. Perform. 25: 1076-96
Brunner, J. S. and Postman, L. (1949) On the perception of incongruity: a paradigm. J. Pers. 18: 206-203.
Bruner, J. S., Postman, L., Rodrigues, J. (1951). Expectation and the Perception of Color. The American Journal of Psychology, 64(2): 216-227.
Bullinaria, John, A. (2007). Understanding the Emergence of Modularity in Neural Systems. Cognitive Science 31: 673-695.
Canavagh, P. (2011). Visual Cognition. Vision Research 51: 1538-1551.
Carruthers, P. (2006). The Architecture of the Mind. Oxford: Oxford University Press.
Cauller, L. (1995). Layer I of primary sensory neocortex: where top-down converges upon bottom-up. Behavioral and Brain Research 71: 163–170.
Cecchi, A. S. (2014). Cognitive penetration, perceptual learning, and neural plasticity. Dialectica 68(1): 63-95.
Churchland, P. M. (1988) Perceptual Plasticity and Theoretical Neutrality: A Reply to Jerry Fodor, Philosophy of Science 55: 167–187.
Courtney, S. M. Petit, L. Haxby, J. V. and Ungerleider, L. G. (1998). The role of prefrontal cortex in working memory: examining the contents of consciousness. Philosophical Transactions of the Royal Society of London B: Biological Sciences 353 (1377): 1819–1828.
Crist, R. E., Li, W. & Gilbert, C. D. (2001). Learning to see: experience and attention in primary visual cortex. Nature Neuroscience 4: 519–525.
Dayan, P., Hinton, G. E., Neal, R. M, Zemel, R.S. (1995). The Helmholtz machine. Neural Computation 7 (5): 889–904.
Deco, G. and Rolls, E. T. (2008). Neural Mechanisms of Visual Memory: A Neurocomputational Perspective. In S. J. Luck and A. Hollingworth (eds), Visual Memory. Oxford: Oxford University Press (pp. 247-289).
Deco, G. and Lee, T.S. (2004). The role of early visual cortex in visual integration: a neural model of recurrent interaction. European Journal of Neuroscience 20: 1089-1100.
Delk, J., Fillenbaum, S. (1965). Differences in perceived color as a function of characteristic color. The American Journal of Psychology 78(2): 290–293.
Davidoff, Jules. (1991). Cognition through Color. Cambridge, MA: MIT Press.
DeRenzi, E. and Spinnler, H. (1967). Impaired Performance on Color Tasks in Patients with Hemispheric Lesions. Cortex, 3: 194-217.
Driesen, N.R. Leung, H-C., Calhoun, V.D., Constable, R.T., Gueorguieva, R., Hoffman, R. Skudlarski, P. Goldman-Rakic, P.S. Krystal, J.H. (2008). Impairment of Working Memory Maintenance and Response in Schizophrenia: Functional Magnetic Resonance Imaging Evidence. Biological Psychiatry 64(12): 1026–1034.
Egan, Frances (1999). “Intentionality and the theory of vision.” In Kathleen Akins (Ed.) Perception. Oxford: Oxford University Press, 232-248.
Farah, Martha. J. (1988). “Is Visual Imagery Really Visual? Overlooked Evidence from Neuropsychology”, Psychological Review, 95: 307-317.
Firestone, C. & Scholl, B. J. (in press). Cognition does not affect perception: Evaluating the evidence for ‘top-down’ effects. Behavioral & Brain Sciences. (http://perception.research.yale.edu/preprints/Firestone-Scholl-BBS.pdf)
Frisby, John P. and Stone James V. (2010). Seeing: The Computational Approach to Biological Vision (2nd edition). Cambridge: MIT Press.
Fodor, J. A. (1983). The Modularity of Mind, Cambridge MA: MIT Press.
Fodor, J. A. (2000) The mind doesn’t work that way: The scope and limits of computational psychology. Cambridge, MA: MIT Press.
Foxe, J. J., Simpson, G. V. (2002). Flow of activation from V1 to frontal cortex in humans: A framework for defining “early” visual processing. Experimental Brain Research 142 (1): 139-50.
Fuster, Joaquin M. (2008). The Prefrontal Cortex. Boston: Academic Press.
Hansen, Thorsten, Olkkonen, Maria, Walter, Sebastian, and Gegenfurtner, Karl R. (2006). Memory Modulates Color Appearance. Nature Neuroscience 9(11): 1367-1368.
Hebart M. N. and Hesselmann, G. (2012). What visual information is processed in the human dorsal stream? The Journal of Neuroscience 32(24): 8107-8109.
Heywood, C. A., Kentridge, R. W. and Cowey, A. (2001). Colour & the Cortex: Wavelength Processing in Cortical Achromatopsia. In B. De Gelder, E. De Haan, & C. A. Heywood (eds.) Varieties of Unconscious Processing: New Findings & Models, (pp. 52-68). Oxford: Oxford University Press.
Heywood, C. A. and Kentridge, R. W. (2003). Achromatopsia, colour vision & cortex. Neurological Clinics of North America 21: 483–500.
Hubel, D. H. and Wiesel, T. N. (1978). Functional architecture of macaque monkey visual cortex. Proc. R. Soc. B (Lond.) 198: 1-59.
Gallese, V. The “conscious” dorsal stream: embodied simulation and its role in space and action conscious awareness. Psyche 13(1).
Gegenfurtner, Karl, Witzel, Christoph, Valkova, Hanna, Hansen, Thorsten (2011) Object Knowledge Modulates Colour Appearance, i-Perception 2: 13-49.
Gilbert, C. D. and Li, W. (2013). Top-down influences on visual processing. Nature Reviews: Neuroscience 14: 351.
Gilbert, C. D. and Li, W. (2012). Adult visual cortical plasticity. Neuron 75: 250–264.
Goldman-Rakic, P. S. (1988). Topography of cognition: parallel distributed networks in primate association cortex. Annual Review of Neuroscience 11: 137–56.
Goldstone, R. L., Braithwaite, D. W., & Byrge, L. A. (In press). Perceptual learning. In N. M. Seel (Ed.), Encyclopedia of the Sciences of Learning. Heidelberg, Germany: Springer Verlag.
Gordon, Ian, E. (2004). Theories of Visual Perception. New York: Psychology Press.
Ito, M. and Gilbert, C. D. (1999) Attention modulates contextual influences in the primary visual cortex of alert monkeys. Neuron 22: 593–604.
Graham, D., Meng, M. (2011). Altered spatial frequency content in paintings by artists with schizophrenia. i-perception 2: 1-9.
Gregory, L. Richard. (2009). Seeing Through Illusions. Oxford: Oxford University Press.
Gregory, L. Richard. (2004). The Blind Leading the Sighted: an Eye-opening Experience of the Wonders of Perception. Nature 430: 1-1, 2004.
Gregory, L. Richard and Jean G. Wallace. (2001). Recovery from Early Blindness—A case study, reproduced from Reproduced from Experimental Psychology Society Monograph No. 2 1963: http://www.richardgregory.org/papers/recovery_blind/contents.htm.
Gregory, L. Richard. (1998). Brainy Mind. British Medical Journal 317: 1693-5.
Gregory, L. Richard. (1997). Knowledge in perception and illusion. Philosophical Transactions of the Royal Society, Series B 352: 1121–1128.
Gregory, L. Richard. (1980). Perceptions as hypotheses. Philosophical Transactions of the Royal Society, Series B 290: 183-97.
Gregory, L. Richard. (1970). The Intelligent Eye. London: Weidenfeld & Nicolson.
Kastner, S., De Weerd, P., Desimone, R. and Ungerleider, L. G. (1998). Mechanisms of directed attention in the human extrastriate cortex as revealed by functional MRI. Science 282: 108–111.
Keane, B. P., Silverstein, S. M., Wang, Y., Papathomas, T. V. (2013). Reduced depth inversion illusions in schizophrenia are state-specific and occur for multiple object types and viewing conditions. Journal of Abnormal Psychology 122(2): 506–512.
Kingdom, F. A. A. (2003). Color brings relief to human vision. Nature Neuroscience 6(6): 641-644.
Kentridge, R., Norman, L. Akins, K., Heywood, C. (2014). Colour Constancy Without Consciousness, presented at the Towards a Science of Consciousness Conference, Tucson, April 2014.
Koethe, D., Kranaster, L., Hoyer, C., Gross, S., Neatby, M. A., Schultze-Lutter, F., Ruhrmann, S., Klosterkötter, J., Hellmich, M., Leweke, F. M. (2009). Binocular depth inversion as a paradigm of reduced visual information processing in prodromal state, antipsychotic-naive and treated schizophrenia. European Archives of Psychiatry and Clinical Neuroscience 259(4): 195–202.
Kottenhoff, H. (1957) Situational and personal influences on space perception with experimental spectacles. Part one: prolonged experiments with inverting glasses. Acta Psychologica 13: 79-97.
Kuehni, Rolf, G. (2003). Olive Green or Chestnut Brown? Behavioral and Brain Sciences 26: 35-36.
Kurson, Robert. (2007). Crushing Through: A True Story of Risk, Adventure, and the Man Who Dared to See. New York: Random House.
Lee, Tai Sing (2003). Computations in the early visual cortex. Journal of Physiology (Paris) 97: 121–139
Lee, T. S., Mumford, D., Romero, R., Lamme, V. A. F. (1998). The Role of the Primary Visual Cortex in Higher Level Vision. Vision Research 38: 2429-2454.
Li, W., Piech, V., Gilbert, C. D. (2004). Perceptual learning and top-down influences in primary visual cortex. Nature Neuroscience 7: 651–657.
Li, W., Piech, V., Gilbert, C. D. (2008). Learning to link visual contours. Neuron 57: 442–451.
Li, W., Piech, V. & Gilbert, C. D. (2008b). Learning to link visual contours. Neuron 57, 442–451.
Mamassian, P., Landy, M., and Maloney, L. T. (2002). Bayesian Modeling of Visual Perception, in Rao, R. P. N., Olshausen, B. & Lewicki M. S. (eds) Probabilistic Models of the Brain: Perception and Neural Function. Cambridge (MA): The MIT Press.
Macpherson, Fiona. (2012). Cognitive Penetration of Colour Experience: Rethinking the Issue in Light of an Indirect Mechanism. Philosophy and Phenomenological Research 74(1): 24-62.
Marr, David. (1982) Vision: A computational investigation into the human representation and processing of visual information. San Francisco, CA: Freeman.
Marr, David. (1976) “Early Processing of Visual Information”, Philosophical Transactions of the Royal Society of London, Series B, Biological Sciences, 275(942): 483-519.
McAlonan, K., Cavanaugh, J. and Wurtz, R. H. (2008). Guarding the gateway to cortex with attention in visual thalamus. Nature 456: 391–394.
McManus, J. N., Li, W. and Gilbert, C. D. (2011). Adaptive shape processing in primary visual cortex. Proceedings of the National Academy of Sciences of the USA 108: 9739–9746.
McRae, K., Jones, M. N. (2013). Semantic Memory. In D. Reisberg (ed.) The Oxford Handbook of Cognitive Psychology (pp. 206-219). Oxford: Oxford University Press.
Milner, A. D. and Goodale, M. A. (2006). The visual brain in action. Oxford: Oxford University Press.
Mitterer, H., Horschig, J., M., Müsseler, J., and Majid, A. (2009). The Influence of Memory on Perception: It’s not what things look like, it’s what you call them. Journal of Experimental Psychology: Learning, Memory, and Cognition 35(6): 1557-1562.
Motter, B. C. (1993). Focal attention produces spatially selective processing in visual cortical areas V1, V2, and V4 in the presence of competing stimuli. Journal of Neurophysiology 70: 909–919.
Mumford, D. (1996). Commentary on ‘Banishing the homunculus’ by H. Barlow. In Knill, D.C. & Richards, W. (eds), Perception as Bayesian. Cambridge: Cambridge University Press (pp. 501–504).
O’Connor, D. H., Fukui, M. M., Pinsk, M. A. & Kastner, S. (2002). Attention modulates responses in the human lateral geniculate nucleus. Nature Neuroscience 5: 1203–1209.
Olkkonen, M., Hansen, T., & Gegenfurtner, K. (2008). Color appearance of familiar objects: Effects of object shape, texture, and illumination changes. Journal of Vision 8(5): 1–16.
Pecher, D., Zwaan, R. A. (2005). Grounding cognition: The role of perception and action in memory, language, and thinking. Cambridge: Cambridge University Press.
Pinto, Y., van der Leij, A. R., Sligte, I. G., Lamme, V. A. F., H. Scholte, S. (2013). Bottom-up and top-down attention are independent. Journal of Vision 13(3): (article) 16.
Pinto, Y., Sligte, I. and V. Lamme (2013a). Working memory requires focal attention, fragile VSTM does not. Journal of Vision 13(9): (article) 459.
Pollen, D. A. (1999). On the Neural Correlates of Visual Perception, Cerebral Cortex 9(1): 4-19.
Pourtois, G., Rauss, K. S. Vuilleumier, P., Schwartz, S. (2008). Effects of Perceptual Learning on Primary Visual Cortex Activity in Humans. Vision research 48: 55–62.
Prinz, Jesse J. (2006). Is the Mind Really Modular? In Robert J. Stainton (Ed.) Contemporary Debates in Cognitive Science. Malden: Blackwell Publishing, pp. 22-36.
Purves D. and Lotto, B. R. (2011). Why we see what we do redux. Sunderland (MA): Sinauer Associates, Inc.
Pylyshyn, Zenon (1999). Is Vision Continuous with Cognition? The Case for Cognitive Impenetrability of Visual Perception, Behavioral and Brain Sciences, 22: 341-423.
Pylyshyn, Z. (1984). Computation and Cognition. Cambridge, MA: MIT Press.
Pylyshyn, Zenon (1973). “What the Mind’s Eye Tells the Mind’s Brain: A Critique of Mental Imagery”, Psychological Bulletin, 80(1): 1-24, 1973.
Raftopoulos, A. (2001). Is Perception Informationally Encapsulated? The Issue of the Theory-Ladenness of Perception, Cognitive Science, 25, 423–51.
Raftopoulos, A. (2009). Cognition and Perception: How Do Psychology and Neural Science Inform Philosophy? Cambridge, MA: MIT Press.
Raftopoulos, A. (2011) Late vision: processes and epistemic status. Frontiers in Psychology 2: (article) 382. doi: 10.3389/fpsyg.2011.00382.
Rao, Rajesh, P. N., Ballard, D. H. (1999). Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nature Neuroscience 2(1): 79–87.
Rauss, K., Schwartz, S. and Pourtois, G. (2011). Top-Down Effects on Early Visual Processing in Humans: A Predictive Coding Framework. Neuroscience and Biobehavioral Reviews 35: 1237–1253.
Rescorla, M. (2013). Bayesian Perceptual Psychology. In Mohan Matthen (ed.)The Oxford Handbook of the Philosophy of Perception.
Riesenhuber, M., Poggio, T. (1999). Hierarchical models of object recognition in cortex. Nature Neuroscience 2: 1019–1025.
Roelfsema, P. R., van Ooyen, A., and Watanabe, T. (2010). Perceptual learning rules based on reinforcers and attention. Trends in cognitive sciences 14(2), 64–71.
Rock, I. (1983) The logic of perception. Cambridge (MA): IT Press.
Sakuraba, S., Sakai, S., Yamanaka, M., Yokosawa, K., Hirayama, K. (2012). Does the human dorsal stream really process a category for tools? The Journal of Neuroscience 32: 3949–3953.
Schacter, D. L. (1987) Implicit memory: history and current status. Journal of Experimental Psychology: Learning, Memory, and Cognition 13: 501-518.
Schlack, A., Albright, T. D. (2007) Remembering visual motion: neural correlates of associative plasticity and motion recall in cortical area MT. Neuron 53: 881–890.
Schneider, U., Borsutzky, M., Seifert, J., Leweke, F.M., Huber, T.J., Rollnik J.D., Emrich, H.M., (2002). Reduced binocular depth inversion in schizophrenic patients. Schizophrenia Research 53: 101–108.
Schneider, U., Leweke, F.M., Sternemann, U., Weber, M.M., Emrich, H.M., (1996). Visual 3D illusion: a systems-theoretical approach to psychosis. European Archives of Psychiatry and Clinical Neuroscience 246: 256–260.
Schoenfeld, M. A., Tempelmann, C., Martinez, A., Hopf, J. M., Sattler, C., Heinze, H. J. & Hillyard, S. A. (2003). Dynamics of features binding during object-selective attention. Proceedings of the National Academy of Sciences of the USA 100: 11806 –11811.
Schroeder, C. E., Mehta, A. D. & Givre, S. J. (1998). A spatiotemporal profile of visual system activation revealed by current source density analysis in the awake macaque. Cerebral Cortex 8: 575–592.
Schwartz, S. (2007). Functional MRI Evidence for Neural Plasticity at Early Stages of Visual Processing in Humans. In N. Osaka, I. Rentschler, and I. Biederman (eds.) Object Recognition, Attention, and Action, pp. 27–40. Tokyo: Springer.
Schwartz, S., Maquet, P. and Frith, C. (2002). Neural Correlates of Perceptual Learning: A Functional MRI Study of Visual Texture Discrimination. Proceedings of the National Academy of Sciences 99: 17137–17142.
Schwartz, S., Vuilleumier, P. Hutton, C. Maravita, A. Dolan, R. J., Driver, J. (2005). Attentional Load and Sensory Competition in Human Vision: Modulation of fMRI Responses by Load at Fixation During Task-Irrelevant Stimulation in the Peripheral Visual Field. Cerebral cortex 15: 770–786.
Siegel, S. (In Press). Rational Evaluability and Perceptual Farce. In J. Zeimbekis and A. Raftopoulos (eds) Cognitive Effects on Perception: New Philosophical Perspectives.
Siegel, S. (2012). Cognitive Penetrability and Perceptual Justification. Noûs, 46(2): 201-222.
Siegel, S. (2010). The Contents of Visual Experience. New York: Oxford University Press.
Siegel, S. (2005). Which properties are represented in perception? In T. Szabo-Gendler & J. Hawthorne (eds.), Perceptual experience (pp. 481–503). Oxford: Oxford University Press.
Sinha, P. and Poggio, T. (2002). Higher-level learning of early visual tasks. In M. Fahle and Poggio T. (Eds.) Perceptual Learning, pp. 273-298. Cambridge (MA): MIT Press.
Siple, P. and Springer, R. M. (1983). Memory and Preference for the Colors of Objects. Perception and Psychophysics 34(4): 363-370.
Spruit, Leen. (2008). Renaissance Views of Active Perception. In Simo Knuuttila and Pekka Kärkkäinen (eds)Theories of Perception in Medieval and Early Modern Philosophy (pp. 203-224). New York: Springer.
Sterzer, P., Haynes, J. D., Rees, G. (2008). Fine-scale activity patterns in high-level visual areas en- code the category of invisible objects. Journal of Vision 8: 1–12.
Styles, E. A. (2006). The Psychology of Attention. Hove: Psychology Press.
Tomasi C. (2001). Early vision, Encyclopedia of Cognitive Sciences 20. London: Nature Publishing Group, Macmillan Reference Limited.
Tulving, E. (1972). Episodic and semantic memory. In E. Tulving & W. Donaldson (eds), Organization of Memory (pp. 381-403). New York: Academic Press.
Tversky, A., Kahneman, D. (1974). Judgment under uncertainty: Heuristics and biases. Science 185: 1124–1131.
Tye, Michael. (2000). Consciousness, Color, and Content. Cambridge (MA): MIT Press.
Ullman, S. (2007). Object recognition and segmentation by a fragment-based hierarchy. Trends in Cognitive Sciences 11: 58–64.
Vallortigara, G. (1999). Segregation and integration of information among visual modules. Behavioral and Brain Sciences 22(3): 398.
Wallbott, H.G., Ricci-Bitti, P. (1993). Decoders’ processing of emotional facial expression—a top-down or bottom-up mechanism? European Journal of Social Psychology 23: 427–443.
Wallis, G. and Bülthoff, H. (2002). Learning to recognize objects. In M. Fahle and Poggio T. (Eds.) Perceptual Learning, pp. 299-316. Cambridge (MA): MIT Press.
Worden, M. S. and Foxe, J. J. (2003). The dynamics of the spread of selective visual attention. Proceedings of the National Academy of Sciences of the USA 100(21): 11933-11935.
Wu, W. (2013). Visual Spatial Constancy and Modularity: Does Intention Penetrate Vision? Philosophical Studies, 165: 647-669.
Yasuda, M., Banno, T. and Komatsu, H. (2010). Color Selectivity of Neurons in the Posterior Inferior Temporal Cortex of the Macaque Monkey. Cerebral Cortex 20(7): 1630-1646.
Zanto, T. P. and Gazzaley, A. (2013). Fronto-parietal network: flexible hub of cognitive connectivity. Trends in Cognitive Sciences 17(12): 602-603.
Zeimbekis, John. (2013). Color and Cognitive Penetrability. Philosophical Studies 165: 167-175.
Zenger, B. and Sagi, D. (2002). Plasticity of low-level visual networks. In M. Fahle and Poggio T. (Eds.) Perceptual Learning, pp. 177-196. Cambridge (MA): MIT Press.
Zhang, N. R. & von der Heydt, R. (2010). Analysis of the context integration mechanisms underlying figure–ground organization in the visual cortex. J. Neurosci. 30: 6482–6496
Zipser, K., Lamme, V. A. & Schiller, P. H. (1996). Contextual modulation in primary visual cortex. Journal of Neuroscience 16: 7376–7389.
Notes
[1] Traditionally, the ventral stream is viewed as the vision-for-perception pathway while the dorsal stream is viewed as the vision-for-action pathway (see Milner and Goodale, 2006). While the ventral stream subserves recognition and discrimination of shapes and objects, the dorsal stream subserves visually guided action, e.g., reaching and grasping based on the moment-to-moment analysis of spatial location, shape, and orientation of objects (see Milner and Goodale, 2006; Hebart and Hesselmaan, 2012). Taken together, the ventral-dorsal visual regions (taken together) are associated with the visual-motor system. Contrary to this traditional view, Gallese (2007) discusses various studies illustrating that motor knowledge is employed to solve perceptual tasks and argues that the dorsal stream is not only responsible for the unconscious control of action but also for the conscious awareness of space and action. In addition, the findings by Sakuraba et al. (2012) are hard to reconcile with the idea that the dorsal stream processes information in the absence of perceptual awareness and, hence, without input from high-level visual areas in the ventral stream (see also Sterzer et al., 2008).
[2] Some hierarchical models suggest that visual perception begins with the analysis of simple features such as orientation and culminating with complex aspects of the visual scene such as shapes and objects (see Marr, 1982; Riesenhuber and Poggio, 1999).
[3] In other words, top-down signals can impart different meanings about the same visual scene based on the behavioral context (Frith and Dolan, 1997; Gilbert and Li, 2013; Purves and Lotto, 2011; Dayan et al., 1995; Pollen, 1999; Lee et al., 1998; Rao et al., 1999; O’Connor et al., 2002; McAlonan et al., 2008). For example, contextual influences (that is, the ways in which the perceptual qualities of a local feature are affected by surrounding scene elements and the way in which characteristics of the global scene affect neuron responses to local features) play an important role in perceptual grouping, perceptual constancies, contour integration, surface segmentation, and shape recognition.
[4] This view is often called constructivism (see Gregory, 1970, 2001, and 2009; Frith and Dolan, 1997; Palmer, 1999; Mamassian et al., 2002; Rescola, 2013).
[5] Interestingly, the relevant memory representations are activated when they touch familiar objects possibly because their tactile perceptual system exhibits the sort of top-down modulation the visual system of normal subjects exhibits in relation to visual perception.
[6] The central tenet of Marr’s hierarchical model is that visual processing involves low (primal sketch), intermediate (2.5-D sketch), and high (3-D model representation) levels, roughly corresponding to different areas of the visual cortex. Accordingly, the function of the primal sketch is to take raw intensity values present in the retinal image and makes explicit information required to detect surfaces; the function of 2.5-D sketch is to makes explicit the orientation and approximate depth of surfaces in a viewer-centered frame of reference—it is viewer-centered in the sense that the emerging image is not yet linked to the external environment but it is organized with reference only to the viewer; and the function of the 3-D model representation is to make explicit shapes and their spatial organization in an object-centered frame of reference as seen by the perceiver—it is object-centered in the sense that the emerging image is independent of particular positions and orientations of the retina. It is only at this high-level of computational processing that the viewer attains a representation of the distal world.
[7] Raftopoulos (2011) uses the term “late vision” to refer to the conceptually modulated stage of visual processing starting at 150-200 ms post-stimulus onset, which, unlike early vision, he considers it to be cognitively penetrable.
[8] Marr offered two reasons for this claim: that visual ‘forms’ can be extracted from the image by using knowledge-free techniques, and that the top-down flowing of information does not affect the computation of the primal sketch.
[9] Raftopoulos allows that late vision, which takes place after the first 120 ms post-stimulus onset, is cognitively penetrable. However, since late vision involves post-perceptual processes, the fact that it is cognitively penetrable does not threaten the CIT (Raftopoulos, 2011).
[10] More recently, it has also been formulated as a causal thesis, according to which cognitive states cannot cause chances in the content or phenomenal character of visual experience (Siegel, 2005, 2012; Stokes, 2013). For example, your belief that the ripe banana in front of you is green cannot cause you to experience it as green. I discuss one of these theses in what follows.
[12] Chromatic variations arise from surfaces such as flowers or painted objects while pure or near-pure luminance variations arise mainly from inhomogeneous illumination such as shadows or shading.
[13] The term ‘colour memory’ refers to a back-end effect in relation to successive colour constancy; it refers to the process used to account for changes in illumination. Specifically, the visual system records the spectral properties of a scene in memory. These mental representations can be activated at a later time to recognize object. I discuss these back-end effects in relation to successive colour constancy below.
[14] Colour constancy was initially attributed to adaptation mechanisms. It was thought that the visual system adjusts its sensitivity to the light according to the context in which the light appears (see Burnhan et al., 1957; Jameson and Hurvich, 1989; Webster and Mollon, 1995). However, recent studies attribute colour constancy to local and global contrast mechanisms (see Kraft and Brainard, 1999; Heywood et al., 2001; Heywood and Kentridge, 2003).
[15] Studies show that although simultaneous colour constancy is limited and variable, successive colour constancy is fast and reliable (Foster and Nascimento,1994).
[16] It is also possible that successive colour contrast mechanisms accounted for these differences between characteristically red figures and the controls. The fact that the scene was lit using a fluorescent light (which has a greenish tint, at least in photographs in the absence of colour correction) may have contributed to the error since it may have had a different effect on the coloured light that was used to generate the background than it had on the surface colour of the figure, thereby creating an ambiguity similar to that causing the public controversy over the blue-black dress. I am grateful to Bob Kentridge for suggesting this possibility.

1. Intro
Dimitria Gatzia’s paper provides helpful clarifications to debates about whether perception is cognitively impenetrable. Gatzia argues that the effects of information stored in high-level perceptual systems (call it “high-level info”) on perceptual processing must be distinguished from effects of cognitive states on perceptual processing. She further argues that the latter, but not the former, count as cognitive penetration. I think this is correct, and is important to keep in mind when searching for evidence of cognitive penetration.
A crucial question going forward is: What distinguishes high-level info from cognitive states? Gatzia writes that cognitive penetration “involves the perceiver’s cognitive states (as opposed to high-level information processing systems of the visual system)” [emphasis in original]. She also contrasts intra-perceptual information with “cognitive states of perceivers (i.e., their beliefs, desires, intentions, which are consciously accessible to the them [sic]).” Passages like these seem to suggest that cognitive penetration involves effects on perception by states that are (i) consciously accessible and (ii) personal-level (rather than subpersonal), while (iii) high-level info is neither consciously accessible nor personal-level, and thus its effects on perception do not count as cognitive penetration. In that case, determining whether the effect of a state is cognitive penetration requires determining whether the state is consciously accessible or personal-level.
I don’t mean to attribute this view to Gatzia. She might not in fact endorse (i)-(iii), or she might endorse them but deny that they account for the difference between high-level info and cognitive states. However, it may be helpful to consider these claims in their own right in the hopes of finding a good metric for distinguishing cognitive states from high-level info. Doing so might help with Gatzia’s project of characterizing cognitive penetration, and with larger projects of mapping out the architecture of the mind.
2. Consciousness
First, whether the state can become conscious is not a promising metric for at least two reasons.
(a) While it may be correct that high-level info is (at least typically) not accessible to consciousness, it’s not clear that this fact plays a role in explaining why top-down effects of high-level info fail to constitute cognitive penetration.
Suppose Phil is a mental duplicate of Lil, except while Lil’s high-level info is unconscious, Phil’s is conscious. We can stipulate that Phil and Lil’s high-level info is alike in all other relevant respects—i.e., both are encapsulated, have the same content, the same representational format, are accessed by the visual system in the same way, and cannot be used directly for cognitive processes (excluding consciousness, if consciousness is cognitive). If a state’s accessibility to consciousness were sufficient for its influence on perception to count as cognitive penetration, then the perceptual states delivered by Phil’s visual system would be the result of cognitive penetration while those delivered by Lil’s visual system would not. But both intuitively and from the perspective of mental architecture, both cases seem to be on a par; there doesn’t seem to be anything fundamentally different about the nature of the processes, such that one should and the other shouldn’t count as cognitive penetration. The fact that a state is conscious, therefore, doesn’t seem to mandate that its influence on perception counts as cognitive penetration.
Perhaps, in normal human minds, when a state is conscious it also plays other cognitive roles, and so perhaps knowing that a state is conscious gives us some reason to think that it is cognitive. But in that case, what makes the state cognitive and what makes its influence on perception count as cognitive penetration is not consciousness itself, but rather some other aspect of mental architecture that correlates with consciousness. In that case, conscious accessibility can provide evidence that a state is cognitive, but is not part of what makes it the case that it is cognitive.
One major caveat: Views like Global Workspace Theory (Baars 1988; Dehaene & Naccache 2001) identify consciousness with certain kinds of cognitive functioning, which might make the above thought experiment incoherent since it stipulates that Phil’s conscious high-level info is functionally equivalent to Lil’s unconscious high-level info. But in that case, consciousness is only a good metric for distinguishing high-level info from cognitive state if we assume certain tendentious theories of consciousness.
(b) Using accessibility to consciousness as a metric for deciding whether a state can cognitively penetrate perception seems to rule out the idea that there are consciously inaccessible but personal-level cognitive states. Take implicit bias. Implicit biases are cognitive states that drive behavior and are arguably attributable to people rather than mere subsystems of people. For instance, if a person crosses the street to avoid someone of a different race because of an implicit bias, that behavior should arguably be explained because of that person’s biases, even if they take the form of consciously inaccessible implicit attitudes. Implicit biases also arguably exhibit the same sort of propositional structure as other paradigmatically cognitive propositional attitudes (Mandelbaum 2016). If an implicit bias directly affects perceptual processing—for example, if it causes someone to perceive an African-American face with a neutral expression as angry—that seems to be a case of cognitive penetration. This seems true even if implicit biases are inaccessible to consciousness (which is, however, a matter of controversy—see, e.g., Hahn et al. 2014).
All of these claims about implicit bias are controversial, to be sure. But we may not want to draw an architectural border between high-level info and information stored in cognition in a way that assumes implicit bias is consciously accessible, or that inaccessible attitudes are never personal-level cognitive states.
Furthermore, repressed beliefs and desires (if they exist) provide another example of cognitive states that are arguably personal-level but not consciously accessible. If someone’s repressed beliefs or desires alter their perceptual processing, that seems to count as cognitive penetration even though the states are not accessible to consciousness.
If these points are correct, then a state’s being consciously accessible is neither necessary nor sufficient for its influence on perception to constitute cognitive penetration.
3. Personal/subpersonal
Second, whether the state is personal-level is not a promising metric either.
To use a toy example, suppose (1) that there is a cheater-detection module (e.g., Cosmides et al. 2010), (2) that it houses subpersonal token propositional states in an information store it uses for typical computations, and (3) that they are accessed by the perceptual system, under rare conditions, in a way that changes the content of the percept in a semantically coherent way. (NB: (3) doesn’t count as a violation of the encapsulation of the cheater-detection module, since it only stipulates that the states are accessed by perception, not that they’re modified by perception.) Why would this not count as a case of cognitive penetration? It strikes me that it would, since information stored in central cognition is being accessed by perceptual systems in a way that alters the content of the percept in a semantically coherent way. Or consider implicit bias again. Even if implicit biases should be understood as subpersonal in addition to being inaccessible to consciousness, it still would seem that any direct effect they have on perceptual processing should count as cognitive penetration.
4. Conclusion
It seems that what matters in deciding whether a state’s influence on perception constitutes cognitive penetration is not whether the state is consciously accessible or personal-level, but rather whether it is a genuinely cognitive, non-perceptual state. The division between high-level info and cognitive states might, therefore, be drawn by appeal to informational encapsulation, representational format (though see Quilty-Dunn 2016), use in proprietarily perceptual algorithms, or some other metric. Gatzia’s paper makes it clear why this division is crucial for understanding cognitive penetration and mental architecture more generally.
References
Baars, B.J. (1988). A Cognitive Theory of Consciousness. New York: Cambridge University Press.
Cosmides, L., Barrett, H. C., & Tooby, J. (2010). Adaptive specializations, social exchange, and the evolution of human intelligence. PNAS 107(2), 9007–9014.
Dehaene, S. & Naccache, L. (2001). Towards a cognitive neuroscience of consciousness: Basic evidence and a workspace framework. Cognition 79, 1–37.
Hahn, A., Judd, C.M., Hirsh, H.K., & Blair, I.V. (2014). Awareness of implicit attitudes. Journal of Experimental Psychology: General 143(3), 1369–1392.
Mandelbaum, E. (2016). Attitude, inference, association: On the propositional structure of implicit bias. Noûs 50(3), 629–658.
Quilty-Dunn, J. (2016). Iconicity and the format of perception. Journal of Consciousness Studies 23(3-4), 255–263.
Dimitria Electra Gatzia’s paper offers a dialectically rich defense of the claim that cognition does not influence (or penetrate) perception, when the key terms are properly understood. While I cannot do justice to all the subtleties of her arguments, my aim is to explore some of the key distinctions Gatzia relies on.
In defending the claim that perception is impenetrable to cognitive influence, Gatzia relies on two contrasts – one between modulation and penetration, and another between the semantic and the causal version of the impenetrability claim.
Gatzia reserves the term ‘cognitive penetration’ for influence on the content and character of perception by other person-level mental states (often described as ‘cognitive’ even though emotions, moods, desires, and other non-belief states, not usually thought of as cognitive outside of this debate, are included). That is, cognitive penetration is a form of influence from one person-level state (a cognitive one like a belief or a desire) to another (a perceptual experience). By contrast, ‘top-down modulation’ is a form of influence from one stage of the visual processing (e.g. V4) to another, earlier or lower stage of visual processing (e.g. V1).
Gatzia grants that there is plenty of top-down modulation in vision, where one stage of visual processing influences another, but no cognitive penetration, where one person-level cognitive state influences the perceptual experience. To make this point stronger, Gatzia emphasizes that she defends the semantic version of the impenetrability claim, which focuses on the contents of the relevant mental states – the purportedly penetrating cognitive state and the perceptual experience. For the semantic impenetrability claim to be true, there must be no influence from the content of cognitive state to the content of the perceptual experience such that there is a match (or ‘semantic coherence’) between those two contents.
The semantic version of the impenetrability claim is to be contrasted with the increasingly popular causal version. The causal version turns on there (not) being some form of causal influence between the cognitive state and the perceptual state, whether or not there is an exact match between the two contents in the end. So the causal version of the impenetrability thesis would be stronger, or more encompassing, claiming that there is no influence at all between the (person-level) cognitive state and the (person-level) perceptual experience, even of the sort that does not yield an exact match between the contents of the cognitive state and the perceptual experience.
Gatzia defends the semantic version of the cognitive impenetrability claim by arguing that some of the currently popular examples of cognitive penetration – particularly those pertaining to color perception – are either cases of top-down modulation rather than penetration, or they are cases of causal influence rather than semantic influence. In assessing Gatzia’s argument and her success in defending the impenetrability claim, I’d like to keep focusing on the two crucial contrasts above – between top-down modulation and cognitive penetration, and between the semantic and the causal version of the impenetrability claim.
First, consider Gatzia’s contrast between top-down modulation and cognitive penetration. It is certainly conceptually useful to draw a line between influence within the perceptual process (modulation) on the one hand and influence from the outside of the perceptual process (cognitive penetration) on the other. But this is also quite the burden to take on, especially if top-down modulation is admitted to be commonplace. After all, Gatzia grants that perception is susceptible to and indeed requires top-down modulation. She only disputes the presence (or perhaps even the possibility) of cognitive influence from the outside of the perceptual process. But to maintain this position, there needs to be a clear and sharp contrast between cognition and perception, so that we can assess which purported forms of influence are from within the perceptual process (top-down modulation) and which are from without (cognitive penetration).
Indeed, by granting that there is top-down modulation, Gatzia grants that perception is not a purely bottom-up process but at least in part a top-down one. If the top-down nature of perception is not in dispute, what exactly is the import of the impenetrability claim? Not that perception can be influenced by high-level states – that much is granted by allowing for the possibility of top-down modulation – but that those influencing high level states are not cognitive. To some, this assessment may bring relief, since all that needs to be established is a firm perception-cognition contrast. But to others, this may bring disappointment, since the debate concerning the possibility of cognitive penetration is meant to enlighten us about these very issues, namely, the boundary and the (top-down or bottom-up) nature of perception, rather than presuppose them to be resolved.
Second, it is unclear why the causal version of the impenetrability claim should be set aside in this debate. Even though Gatzia is only interested in influence on perception by person-level content-bearing mental states, that influence could be exerted via intermediary sub-personal information-bearing stages. At levels below personal, there is no mental content to speak of, which seemingly makes semantic influence impossible. But that does not mean that cognitive person-level states could not first exert their influence downward, towards some information-processing stage, and then back up to person-level perceptual experience. In short, person-level cognitive influence could be mediated by sub-personal level processing, rather than influencing the perceptual experience directly. (In fact, perceptual learning sometimes seems to happen in just this way.)
If cognitive states can influence perceptual experience indirectly, via intermediately stages, then the causal impenetrability claim is just as relevant as the semantic version. After all, information-bearing states, rather than content-bearing ones, can only be influenced causally, not semantically, so the causal version of the impenetrability claim seems to be tied up with the semantic one. Perhaps Gatzia means to rule out that cognitive states could exert their influence on perceptual experience indirectly as sketched, in which case I would wonder why.
In sum, Gatzia’s argument for the impenetrability of perception to cognitive influence relies on some valuable contrasts – between top-down modulation and cognitive penetration, as well as between the semantic and the causal version of the impenetrability claim – both of which are worth keeping in mind, no matter which side of the debate one occupies. However, I suspect that those contrasts are harder to draw clearly than Gatzia leads on. Moreover, favoring the semantic over the causal version of the impenetrability claim artificially restricts the debate about the possibility of cognitive penetration.
Perceiving facial emotions: late visual effects?
I
In her paper “Cognitive penetration and top-down modulation in visual perception,” Dimitria Electra Gatzia argues against the cognitive penetration of early vision in two cases: the visual perception of facial emotions (Butler et al. 2008, Carroll and Russell 1996) and color perception (Delk and Fillenbaum 1965, Hansen et al. 2006). First I’ll assess Gatzia’s account of the experiments on facial emotions and color perception (§I). Then I’ll describe what I take to be a fault in interpretations of the findings on facial emotions, namely: the failure to distinguish cognitive effects on early vision from effects on late vision (§II).
(1) Gatzia’s account of the facial emotion experiments. In the experiments by Butler et al. (2008), subjects perceived the same facial expression (E) as angry or as fearful depending on which facial expression (conversely, fearful or angry) they were exposed to immediately prior to perceiving E. This is thought to be an effect of perceptual adaptation: the neural firing thresholds for detecting fear are raised by perceiving a fearful face, so if we perceive a less fearful face immediately afterwards, vision is less apt to detect fear. If the new facial configuration is equally consistent with fear and anger, then the subject is more likely to detect anger than to detect fear. In the experiments by Carroll and Russell (1996), facial emotion recognition is not influenced by a perceptual mechanism, as in the previous case, but by priming. Subjects perceive a face that expresses one emotion (eg, fear) as expressing another emotion (anger) because prior to perception they have heard a narrative which primes the second emotion.
D.E. Gatzia is interested in whether these findings amount to forms of cognitive penetration of perception, so she targets an interpretation of the findings by Marchi and Newen (2015). On that interpretation, functioning of the visual mechanisms that generate perceptual experiences of faces is affected by the activation of conceptual representations. The result is that those visual mechanisms yield different visual experiences of faces depending on the conceptual input they receive. Gatzia argues that Marchi and Newen’s analysis falls foul of an interpretive pitfall described by Firestone and Scholl (2015): it misinterprets cognitive effects on memory access as cognitive effects on perception.
Firestone and Scholl describe this error at work in interpretations of experiments where subjects are asked to recognize stimuli at very short exposures, a task whose outcome clearly depends on memory access. For example, Gantman and Van Bavel (2014) claim that at exposure times of 40-60 ms, subjects have greater facility in recognizing morally charged words than morally neutral ones. Gantman and Van Bavel take this to mean that semantic (cognitive) information influences very early visual information processing. But Firestone and Scholl point out that in the course of the experiments, subjects’ memories of morally charged words are gradually activated prior to stimulus onset; this would speed up the matching of templates to percepts, and thus recognition, but without affecting early visual processing that yields those percepts.
How relevant is this pitfall (“pitfall # 6” in Firestone and Scholl’s compendium) for the experiments on facial emotions? First, in Butler et al.’s adaptation experiments, any working memory of the adaptor stimulus (while perceiving the test stimulus) would have the opposite effect to priming. For instance, if the adaptor stimulus was a fearful face, it would cause subjects to classify the test stimulus not as fearful—as one would expect if priming were at work—but as angry. So priming of memory access cannot explain the outcome of those experiments.
Secondly, neither Butler et al. (2008) nor Carroll and Russell (1996) use very short exposure times, because their experiments are not intended to measure facility or speed of recognition. Instead, the task is to classify a stimulus exposed for 300 ms, which suffices for conscious recognition to occur during exposure. Thus, priming of memory access—which facilitates feedback during unconscious template matching by activating suitable memorized representations in advance—isn’t required for the subject to recognize the stimulus as either fearful or angry. It’s not that, for lack of time, memories cannot be accessed quickly enough for the stimulus to be classified, or that classification has to fall back on the primed concept as the only one available in the time frame. It’s that although subjects have ample time to access any relevant memory representations required for unconscious template matching, in one experiment they systematically place ambiguous stimuli under only one class, while in the other they mis-classify the facial emotion altogether.
Therefore, priming of memory access doesn’t seem to be a promising explanation of these two sets of findings on facial emotions. On the other hand, I share Gatzia’s scepticism about using the findings to conclude in favor of the cognitive penetration of early vision. So I’ll return to the facial emotion experiments in §II, where I’ll ask whether the conclusion could be denied on the grounds that only late visual states are affected by priming.
(2) Gatzia’s account of the color perception experiments. I turn now to D. E. Gatzia’s argument against cognitive penetration in color perception. That argument begins in sections 3 and 4 of her paper, where she distinguishes the cognitive penetration of vision from top-down modulation of vision, and runs to the end of section 5 where she discusses the classic experiments by Hansen et al. (2006) and Delk and Fillenbaum (1965).
The structure of the argument is as follows. (1) There are forms of top-down modulation of early vision which do not amount to cognitive penetration, namely, hard-wired visual processing principles. (2) Color constancy is such a form of top-down modulation of early vision. (3) The findings of Hansen et al. (2006) and Delk and Fillenbaum (1965) can be explained as effects of color constancy. (4) Therefore, the findings do not show cognitive penetration of vision.
Premise (1) should be granted, it’s a widely acknowledged point and has played a key role from the beginning in the literature on penetration, encapsulation and modularity. For that reason, perhaps the point shouldn’t be presented as novel, in the way it is here. For example, Gatzia writes (p. 8) that Raftopoulos (2009) “maintains that early vision … is impervious to top-down modulation”; whereas in fact Raftopoulos (2009) describes several forms of local top-down processing in early vision, taking pains to explain why they don’t count as cognitive penetration. Generally, the very concepts of a processing hierarchy in Marr (1982), encapsulated modules in Fodor (1983), and early vision in Pylyshyn (1999, 2003) assume that, at a certain level of description, early vision appears to embody a form of knowledge and to perform inferences. But all of those authors have argued in one sense or another in favour of the encapsulation of early vision, and consistently with that, none of them is claiming that in early vision personal-level beliefs or sentence-like generalizations are applied to process information.
The key step in Gatzia’s argument is (3), namely, that the findings of Hansen et al. (2006) and Delk and Fillenbaum (1965) “can be explained in terms of successive colour constancy”, in which “we see the same object as having the same colour under different illuminations” (p. 13). An obstacle to adopting this view is that in those experiments, responses are specific to recognitional concepts for kinds, so they can’t be attributed to hard-wired constancy mechanisms which are dedicated to computing shape from illumination as a requisite for triggering recognitional concepts. Gatzia acknowledges this obstacle, but then holds that color constancy effects occur as a result of priming: “the relevant representations in memory are only activated for the characteristically red objects but not the controls making the visual system prone to error”. But in that case, constancy cannot be a hard-wired visual formation principle immune to cognitive influence: the triggering of the recognitional concept by the stimulus affects how color constancy mechanisms work, making the color-associated objects appear a different color to the neutral objects. So if Gatzia wants to maintain point (3) above (that the findings are explained by color constancy), then she has to drop point (2) (that color constancy is a hard-wired process immune to cognitive influence), and cannot conclude that the findings do not show cognitive penetration of vision.
Finally, drawing on the hard-wired nature of color constancy mechanisms, Gatzia argues that “these results do not support the claim that cognitive penetration occurred since these subjects would be unable to alter computations they have no conscious awareness of in such a way as to alter their visual experience.” This is a petitio principii: it argues that there’s no cognitive penetration of early vision in the particular experiments because early vision is cognitively impenetrable. In fact, what Gatzia describes would be a textbook case of cognitive penetration of early vision: information in cognitive states (long-term memory, concepts for kinds) affects low-level visual processing (color constancy mechanisms) to yield a visual experience counterfactually dependent on the cognitive information, and the visual experience is used in an epistemically damaging way to base false color judgments.
I’m sympathetic to Gatzia’s view that the findings on color perception don’t support cognitive penetration of early vision. It’s just that I don’t think they can be explained as effects of color constancy, at least on the basis of what Gatzia writes here. However, two proposals have already been put forward about the same experimental findings denying that they imply cognitive penetration. One explains them as effects on judgment instead of vision (Zeimbekis 2013); the other claims that they’re due to early visual shape-colour associations, which would disqualify the effects from being cognitive (Deroy 2013).
II
Also, like Gatzia, I’m sceptical that the facial emotion findings show cognitive penetration of early vision. But if my objections to her view are right, then cognitive penetration can’t be avoided by imputing the subjects’ responses in the experiments to primed memory access. Is there any other way to explain the findings while also maintaining the encapsulation of early vision? My hunch is that in neither of the above experiments on facial emotions is early vision under cognitive influence.
The interpretation of the facial emotion findings that Gatzia seeks to disqualify is Marchi and Newen’s (2015). Marchi and Newen (§3) argue for cognitive penetration in two steps. (1) First, they adopt Block’s (2014) account of adaptation as a perceptual-level phenomenon, with the objective of securing the point that “facial expressions as wholes are perceptually processed” in a process of perceptual integration of “compounds” of low-level features. Cognition is disqualified from playing this role on the grounds that the integration process occurs “before corresponding judgments about the emotion expressed by the faces are formed”. (2) Next, Marchi and Newen claim that this “perceptual integration process of facial expressions may be influenced by contextual background knowledge and the subject’s beliefs” in what qualifies as a form of cognitive penetration of perception.
Point (1) is intended to secure that the states or processes affected by cognition are perceptual states. However, Marchi and Newen don’t say whether it’s early or late visual states that are affected by cognitive states on their account. On current definitions of the cognitive impenetrability of perception, what is encapsulated from cognition is not all of perception, but a subset of visual processes dubbed “early vision” (Pylyshyn 1999; for an overview of the distinction see Raftopoulos and Zeimbekis 2015, §2.2). Therefore, to reach their conclusion about cognitive penetration, Marchi and Newen have to show that early vision is cognitively penetrated. On Pylyshyn’s (1999) and Marr’s (1982) criteria, early vision includes no more than the visual processes that generate the experience of viewer-centered volumetric shape, experienced color, and experienced textures, slants and orientations.
However, when Marchi and Newen (2015) argue that the perception of emotions is cognitively penetrated, they have other visual outputs in mind. Instead of low-level features like shape or orientation, they define the relevant outputs of vision as compounds of low-level features “integrated at the level of perception” and giving rise to specific perceptual experiences. On the question of how different compounds of low-level features could emerge from the same stimulus, Marchi and Newen have this to say: “some relevant perceptual information conveyed by the face is disregarded or given increased saliency. This is precisely what allows two different compounds to be formed” (2015, §5).
To assess this proposal, consider the stimuli used by Butler et al. (2008), such as Figure 2A. The figure consists of several pictures of the same face; the face to the far left is angry (adaptor stimulus), the face to the far right is fearful (adaptor stimulus), and the middle face is a morphed image ambiguous between fear and anger (test stimulus). For the experiment to exploit adaptation, attentional resources have to be focused on the same low-level features (eg orientation for eyebrows, shape for eyelids and lips) when detecting fear and when detecting anger, because adaptation means that the neural codings are altered for those low-level features. Therefore, low-level features are not “disregarded or given increased saliency” compared to other low-level features of the test stimulus as a result of different adaptations, and that is not how “different compounds” can be formed from the same stimulus, contrary to what Marchi and Newen hold. Instead, features are experienced as salient compared to corresponding features in the adaptor stimulus perceived previously. For example, when we shift from the adaptor face to the test face, the orientation of the eyebrows will be saliently higher or saliently lower.
Given that subjects attend to the same low-level features of the same test stimuli irrespective of which emotion they detect, then how could different visual experiences of the same stimulus emerge? How could “different compounds” be “formed” by the perceptual formation process Marchi and Newen describe, when that process functions under the influence of priming in the other set of experiments (Carroll and Russell 1996).
One way for two different experiences to emerge from the same stimulus in Carroll and Russell’s experiments would be this: depending on which concept is primed, vision assigns different low-level features (that is, different shapes, slants or orientations) to the same test-stimulus (that is, to the same eyebrows, eyes, lips, etc). In that case, subjects have illusory visual representations of the low-level properties each time: they don’t see the position of the eyebrows or the shape of the mouth veridically. It seems highly speculative to conclude that this is how different compounds of low-level features emerge from the same stimulus. Marchi and Newen (§5) present their argument as an inference to the best explanation, but this kind of explanation doesn’t pass that test because it makes vision so obedient to concepts that it begins to resemble visualizing and loses its epistemic role as a form of perception.
This problem is compounded by a difference between the two sets of experiments. In the adaptation experiments, Butler et al. used test stimuli ambiguous between anger and fear and asked subjects to classify them: “subjects adapted to one of two adapting stimuli at the extreme ends of a morph continuum, and then were tested for aftereffects on test stimuli selected from the mid-region of the continuum” (Butler et al. 2008, section 2.1). But in Carroll and Russell (1996), test stimuli were not ambiguous; they were protoypical facial expressions of emotions, comparable to Butler et al.’s adaptor stimuli. So for Carroll and Russell’s findings to be explained as cognitive effects on shape and orientation assignment, subjects would have to misperceive low-level features by far greater magnitudes than they (putatively!) do in the adaptation experiments, making vision even more pliable to concepts.
Marchi and Newen’s rejoinder to this type of consideration is that if subjects can form “different judgments on the basis of the same perceptual experience”, then “we do not see how constraints on emotion recognition could be introduced in a principled way” (2015, §5). Well, there is a principled way to constrain recognition generally: by using the cognitive impenetrability hypothesis. On that hypothesis, early vision delivers information about low-level features independently of any influence from conceptual states (although conceptual states like thought or imagining can influence, and even be part of, late visual states). If Marchi and Newen are claiming that early vision is affected in the experiments in the way just outlined—namely, that vision assigns different low-level features to the same test-stimulus depending on which concept has been primed—then they can’t avail of this principled way of constraining recognition. If vision is so pliable that we see objects as having whatever shapes and orientations would be consistent with our thoughts and expectations, then what hope is there of a principled concept of recognition—for emotions or anything else?
But there is another way that two different experiences could emerge from the same stimulus. It could be that subjects have different late visual experiences of the same set of low-level features. The low-level features—essentially, shapes and orientations—are identical each time; the perceptions are part of late vision, and they differ. In this framework, the salience effects Marchi and Newen have in mind could emerge in any of the following ways: (a) they could be experiences of the contrast between test and adaptor stimuli; (b) experiences of the contrast between the test stimulus and a representation of the adaptor stimulus in working memory; or even (c) result from the contrast between test stimulus and recognitional templates for facial emotions which have just been activated by the adaptor stimulus. For any of these forms of salience to emerge, vision has first to assign shape to the test stimulus veridically; if it did not, there would be no salience relative to the previous stimulus. But in that case, the salience effect can’t occur unless vision has already assigned shape. So priming does not affect visual shape and orientation assignment, which are the relevant early visual processes here. The salience effect would then be a case of what Lyons (2011) calls a “late experiential effect leaving the nonconceptual early perceptual states unaffected”, and the early visual processes leading up to shape and orientation assignment would not be cognitively penetrated.
References
Block N. (2014). Seeing-as in the light of vision science. Philosophy and Phenomenological Research 89: 560-572.
Butler A., Oruc I., Fox C. J., Barton J. J. S. (2008). Factors contributing to the adaptation aftereffects of facial expression. Brain Research 1191: 116-126.
Carroll J. M., Russell J. A. (1996). Do facial expressions signal specific emotions? Judging emotions from the face in context. J. Pers. Soc. Psychol. 70: 205-218
Delk, J. L., & Fillenbaum, S. (1965). Differences in perceived colour as a function of characteristic colour. The American Journal of Psychology, 78(2), 290-293.
Deroy, O. (2013). Object-sensitivity versus cognitive penetrability of perception. Philosophical Studies, 162: 87-107.
Firestone, C. and Scholl, B. (2015). Cognition does not affect perception: Evaluating the evidence for ‘top-down’ effects. Forthcoming in Behavioral & Brain Sciences.
Fodor, J. (1983). The Modularity of mind. Cambridge, Mass.: The MIT Press.
Gantman, A. and Van Bavel, J. (2014). The moral pop-out effect: Enhanced perceptual awareness of morally relevant stimuli. Cognition 132: 22-29.
Hansen, T., Olkkonen, M., Walter, S., and Gegenfurtner, K. (2006). Memory modulates colour appearance. Nature Neuroscience, 9(11), 1367–1368.
Lyons, J. C. (2011). Circularity, reliability, and the cognitive penetrability of perception’. Philosophical Issues 21, 289-311.
Marchi, F. and Newen, A. (2015). Cognitive penetrability and emotion recognition in human facial expressions. Front. Psychol. DOI: 10.3389.
Marr, D. (1982). Vision: A Computational Investigation into Human Representation and Processing of Visual Information. Freeman.
Pylyshyn, Z. (1999). Is vision continuous with cognition? Behavioral and Brain Sciences, 22, 341-365.
Pylyshyn, Z. (2003). Seeing and Visualizing: It’s not what you Think. Cambridge, Mass.: The MIT press.
Raftopoulos, A. (2009). Cognition and Perception. Cambridge, Mass.: The MIT Press.
Raftopoulos, Α. and Zeimbekis, J. (2015). Cognitive Penetrability: An Overview. In The Cognitive Penetrability of Perception, Oxford University Press; pp. 1-56.
Zeimbekis, J. (2013). Color and cognitive penetrability. Philosophical Studies 165.1, 2013: 167-175.
Many thanks to the organizers and the commentators!
I will respond to each commentator separately, roughly in the order I received the comments.
I am in agreement with Jake Quilty-Dunn in that “the effects of information stored in high-level perceptual systems (call it “high-level info”) on perceptual processing must be distinguished from effects of cognitive states on perceptual processing.” Indeed, as he notes, the primary focus of my paper is to show that only the latter count as instances of cognitive penetration (henceforth, CP).
Jake asks “What distinguishes high-level info from cognitive states?” and says that my treatment of cognitive states (that is, I treat them as states of perceivers as opposed to merely high-level information of the visual system) suggests that these states seem to be (i) consciously accessible and (ii) personal-level while higher-level perceptual processing lacks both properties. Jake does not attribute this view to me but I agree with him that this is an important discussion to have so I want to say a few things about each of the concerns (a) and (b) he brings up.
With respect to (a), Jake’s worry is as follows: “While it may be correct that high-level info is (at least typically) not accessible to consciousness, it’s not clear that this fact plays a role in explaining why top-down effects of high-level info fail to constitute cognitive penetration.” I do not think that the relevant information (or the way that information is processed) that brings about the relevant effects has to be conscious and that is the reason I have refrained from even suggesting that either it is or it must be. The distinction I am trying to make is between, on the one hand, (i) states of the visual system on which the perceiver’s believes, desires, etc., could not possibly have an effect on (e.g., colour contrast mechanisms, which determine what colours objects appear to have to a perceiver independently of their beliefs (or other cognitive states) and, on the other, (ii) states of the visual system that the perceiver’s believes, desires, etc., could have an effect on (e.g., believing that the banana is green and seeing it as green when it is in fact yellow). The perceiver does not have to be conscious that her cognitive states have an effect on her visual experience in the latter case, not does she have to be conscious of the processing that gives rise to such altered experiences (e.g., of the banana looking green). Although both (i) and (ii) can be construed as top-down influences, it seems to me that only (ii) is relevant to the discussion of CP. While (i) affects how things appear to a perceiver, they could not be described as cases of CP because what produces the effect is not the cognitive states of the perceiver—these are simply states of the visual system that cannot be affected by the cognitive states of the perceiver (again such as colour constancy effects). Nevertheless, neither of these top-down effects (i) or (ii) need be conscious nor do they need to differ in their output. I think that this distinction is very important in part because most of current debate on CP is focused on the former cases. Now, if I am right that the perceivers are not conscious of the influences pertaining to (ii), we have to find some other ways to empirically test whether CP occurs since that would most definitely move the debate forward. So I agree with Jake that how we perceive the world can be influenced by our mental states such as beliefs or desires even though we may be unconscious that such influences are taking place or how they are taking place. However, here too we have to be careful. For example, studies in social sciences seem to suggest that unconscious racist beliefs or other implicit biases can influence our judgments of other people. However, such cases would not count as cases of CP so long as these states affect our judgments of what we visually experience and not our visual experience per se. The idea (at least in Pylyshyan terms) is that cognitive states perceivers are in should be able to alter the function their visual system computes in a way that it can alter their visual experience.
With respect to (b), namely, “Using accessibility to consciousness as a metric for deciding whether a state can cognitively penetrate perception seems to rule out the idea that there are consciously inaccessible but personal-level cognitive states,” my response is roughly the same: it must be the case that it is the cognitive states of perceivers that are able to alter the function their visual system computes in such as way that alters their visual experience. It need not be the case that this information is accessible to consciousness or not. Again, the cognitive states that affect visual perception need not be consciously accessible. Jake suggests that implicit bias is an example of a top-down effect that tends to not be accessible to consciousness. I agree. But I should point out that such cases are typically explained in terms of judgments influencing what we visual experience as opposed to influencing our visual experience per se. Marchi and Newen, for example, go to great lengths to argue that the results of the experiments on facial expressions are not the effects of judgments perceivers make. I think these distinctions are very difficult to make and I would invite more discussion on the topic because therein, I think, lies the key to answering the question of whether visual experience is cognitively penetrable.
* * *
I am also in agreement with John Zeimbekis in that the experiments I discuss do not support the claim that CP occurs, albeit for different reasons. John argues that, in my discussion of facial expressions, I fail to distinguish the effects on early vision from the effects on late vision. I will address his argument in what follows. Let me first emphasize that I am not in agreement with Thanos Raftopoulos who distinguishes between early and late vision. As I argue in the paper, I find his definition of early vision to be problematic (the same goes for the definitions of Marr’s and Pylyshyn’s). Raftopoulos (2011) defines early vision as the processing stage that lasts no more than 100-120 ms post-stimulus onset. (Raftopoulos (2011) uses the term “late vision” to refer to the conceptually modulated stage of visual processing starting at 150-200 ms post-stimulus onset, which, unlike early vision, he considers it to be cognitively penetrable.) However, studies on humans indicate that 100-120 ms is actually a remarkably long time in terms of neural transmission given that many regions of the cortex, including regions of the frontal cortices, can be activated in half that rate. Raftopoulos acknowledges that some such effects occur but he thinks that they are post-perceptual. I disagree with him on this point also. When I wrote that Raftopoulos “maintains that early vision… is impervious to top-down modulation”, as John points out, my remarks were in the context of arguing that any such modulation does not count as perceptual (and hence not as a case of CP) for Raftopoulos. John seems to think that I am attributing another view to Raftopoulos since he goes on to say that “whereas in fact Raftopoulos (2009) describes several forms of local top-down processing in early vision, taking pains to explain why they don’t count as cognitive penetration.” I tried to make it clear in the paper that Raftopoulos allows for such influences but treats them as “post-perceptual” and do not have the view John attributes to me.
John objects to my assessment of Marchi and Newen’s (M&N) argument (for the claim that experiences of facial expressions are cognitively penetrated by cognitive states) on the basis that the case I discuss involves priming. (I should mention here that the point of my argument is to show that the operative definition of CP used by M&N is problematic because it fails to make the distinction that I think is needed if we are going to be able to determine whether CP occurs or not in these cases.) John argues that my assessment of the studies on facial expressions used by M&N show that the results cannot be attributed to a back-end effect on memory access, e.g., activating the relevant representation in memory when a stimulus is present, because these experiments do not use fast exposures and are thus not intended to measure recognition but to classify a stimulus exposed for 300 ms. As a result he writes that “priming of memory access—which facilitates feedback during unconscious template matching by activating suitable memorized representations in advance—isn’t required for the subject to recognize the stimulus as either fearful or angry.” So, according to John, “although subjects have ample time to access any relevant memory representations required for unconscious template matching, in one experiment they systematically place ambiguous stimuli under only one class, while in the other they misclassify the facial emotion altogether.” I suspect that the type of memory John has in mind is either working or long-term memory, which is required to complete a task, whereas, as I understand it, F&S’s discussion regarding this criterion is meant to highlight a distinction between front-end and back-end recognitional effects, where the latter (at least in the case of colour involves colour memory –understood not in the sense that subjects remember the colours of things but rather in the sense that the visual system “remembers” the colours of things as is the case in colour constancy. I think the same can be said for facial expressions. We know that one of the first things our visual system “learns” is to recognize faces. It is therefore plausible that (lets call it) face memory (as a way to bring into attention that they are akin to colour memory) is doing the work, not the memories the perceivers related to priming. It seems to me that unconscious memory representations (resulting from priming that took place earlier in the experiment) are activated by a combination of the visual target and the context – indeed research suggests that different cues are added together when we interpret facial expressions (Wallbott and Ricci-Bitti, 1993)—and this process requires not working or long-term memory but face memory.
John also objects to my argument about how we should interpret the results in the Hansen et al. (2006) experiment. John explains these results in terms of judgments but it seems to me that the results can be more plausibly explained by reference to the effects of colour constancy lest we want to attribute an error in judgment to subjects (see, for example, Macpherson (2012) who offers some good reasons for rejecting this strategy). It seems to me that the fact that subjects adjusted the colour of the banana to give it a bluish tint can be more plausibly explained by the fact that their visual system interpreted the image as being more yellow when it was actually gray because their colour constancy mechanisms have been conditioned (or “learned”) to view bananas in daylight, which consists of a bluish light. We know that repeated exposures result in long-lasting structural or functional changes in the visual system. (Some take these cases to be cases of diachronic CP, see Churchland (1988) and Cecchi (2014), for example, whereas I think they are cases of perceptual learning. I believe the suggestion that cases of perceptual learning are cases of CP highlights the difficulties that arise when we refrain from making the distinction I am trying to make in my paper). The visual system, therefore, “learns” to discount blue light when confronted with scenes that consist of naturally yellow objects. We would expect that such long-lasting structural or functional changes to have an effect, i.e., failure of colour constancy, in experimental circumstances: the visual system must have interpreted the banana’s colour in the way that it would have interpret it under daylight. But that falls under, what in my response to Jake I called, (i) – i.e., states of the visual system on which the perceiver’s believes, desires, etc., could not possibly have an effect on (e.g., colour contrast mechanisms, which determine what colours objects appear to have to a perceiver independently of their beliefs (or other cognitive states) distinction – and so it cannot said to be a case of CP.
Finally, John argues that I am begging the question because, according to him, I argue “that there’s no cognitive penetration of early vision in the particular experiments because early vision is cognitively impenetrable.” He goes on to say that what I describe “would be a textbook case of cognitive penetration of early vision: information in cognitive states (long-term memory, concepts for kinds) affects low-level visual processing (color constancy mechanisms) to yield a visual experience counterfactually dependent on the cognitive information, and the visual experience is used in an epistemically damaging way to base false color judgments.” I offer arguments for my claim that we should reject the distinction between early and late vision. I also do not merely assume that vision is cognitively impenetrable; I argue that the results do not show that it is because they can be explained in terms of colour constancy mechanisms (see also Brogaard and Gatzia, 2016), which do not involve working or long-term memory but colour memory (meaning that the computations associated with colour constancy cannot be altered by the subject). Under Pylyshyn’s definition, which I am using in the paper, that is not a case of CP.
* * *
Katia Samoilova rightly notes that we should make “a clear and sharp contrast between cognition and perception, so that we can assess which purported forms of influence are from within the perceptual process (top-down modulation) and which are from without (cognitive penetration).” This is a task I take in the longer project that this paper fits in. Katia is correct in saying that I deny that perception is purely a bottom-down process – there is a lot of evidence for this claim, which I discuss in the larger project. She asks: “If the top-down nature of perception is not in dispute, what exactly is the import of the impenetrability claim?” What I try to bring attention to is that talking about top-down modulation is insufficient to talk about CP because the latter, at least to me, is supposed to be a question of whether the perceiver’s cognitive states, e.g., beliefs, desires, as opposed to whether the adaptations of her visual system, can influence how she perceives the world. For example, can my belief that the banana is green influence my visual experience in such a way that I come to see it as green? The question is not merely about whether high-level processes within a sense modality can influence low-level processes within the same (or perhaps other) modality (modalities). The question should be about whether perceivers have the ability to alter the computations that are taking place within their visual modality in such as way as to alter their experiences. I think this is an important clarification, one that can move the debate forward. (Part of the reason I leave aside the causal claim is that I think that many of the indirect influences are actually cases of perceptual learning, which I do not count as cases of CP for reasons I cannot go into here but are also part of the larger project.) Katia writes: “I suspect that those contrasts are harder to draw clearly than Gatzia leads on. Moreover, favoring the semantic over the causal version of the impenetrability claim artificially restricts the debate about the possibility of cognitive penetration.” I agree with Katia in that these are very difficult distinctions to make and I hope I have not given the impression that I thought otherwise. Also I agree that we are restricting the possibility of CP by choosing a certain definition, although I cannot see any other way to, at least, begin clarifying the issue. However, as I see it, the bigger issue is not whether we adopt the semantic over the causal thesis (since they are more similar than it may appear) but rather why we should frame the discussion in terms of a computational theory of mind. In the larger project, I attempt to sort out through these issues.
I hope that I have adequately addressed the concerns raised. If I have not, I hope we can continue the conversation during the conference.
Many thanks to Jake, John, and Katia for valuable comments!
Best wishes,
Dimitria Gatzia